In the past, flavor development for e-liquids was a creative pursuit driven by consumer trends. Today, it’s a legal and scientific challenge. With the tightening of international e-cigarette regulations—particularly the European Union’sTobacco Products Directive (TPD)and theU.S. FDA’s Premarket Tobacco Product Application (PMTA)—flavorings are under regulatory scrutiny like never before.
To achieve global market entry and consumer trust,flavor formulations must meet the dual challenge of delivering sensory appeal while remaining fully compliantwith evolving legislation.
TheTPD (Directive 2014/40/EU)doesn’t just govern nicotine levels or packaging—it regulates what you can and cannot put into the flavoring component of e-liquids:
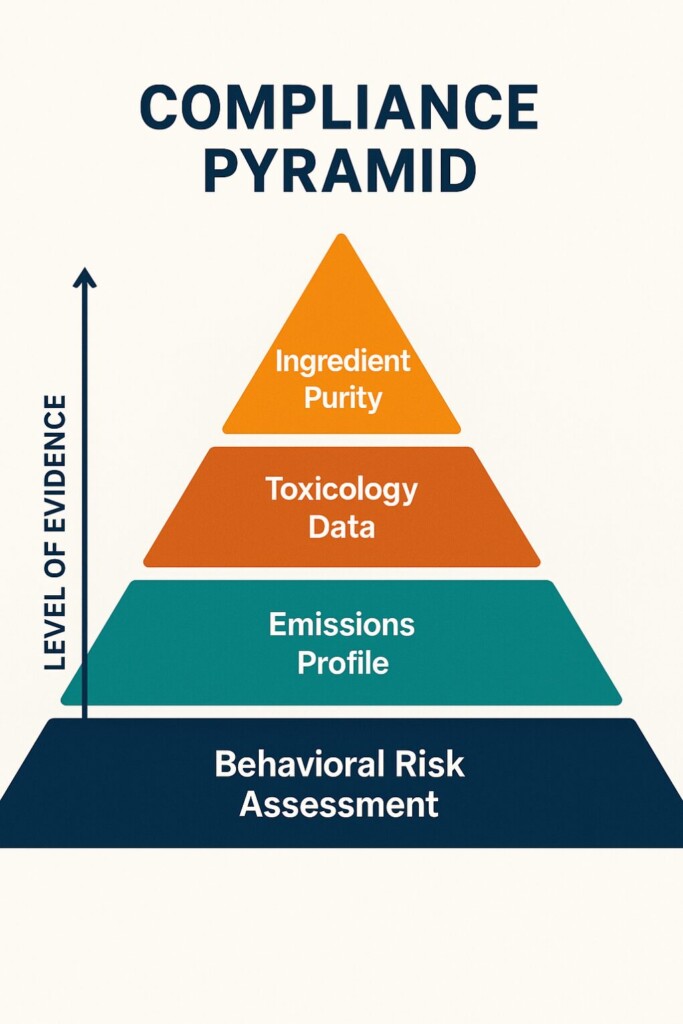
Under thePMTA, manufacturers must providescientific evidencethat the flavoring used is“appropriate for the protection of public health.”This involves:
Even experienced formulators may unintentionally fail compliance tests. Let’s highlight some frequent mistakes:
Using unapproved carriers likeTriacetin, Diacetyl, oderBenzaldehydein excess—even if flavor-enhancing—can trigger bans.
“Natural” doesn’t equal “safe.” Extracts must be:
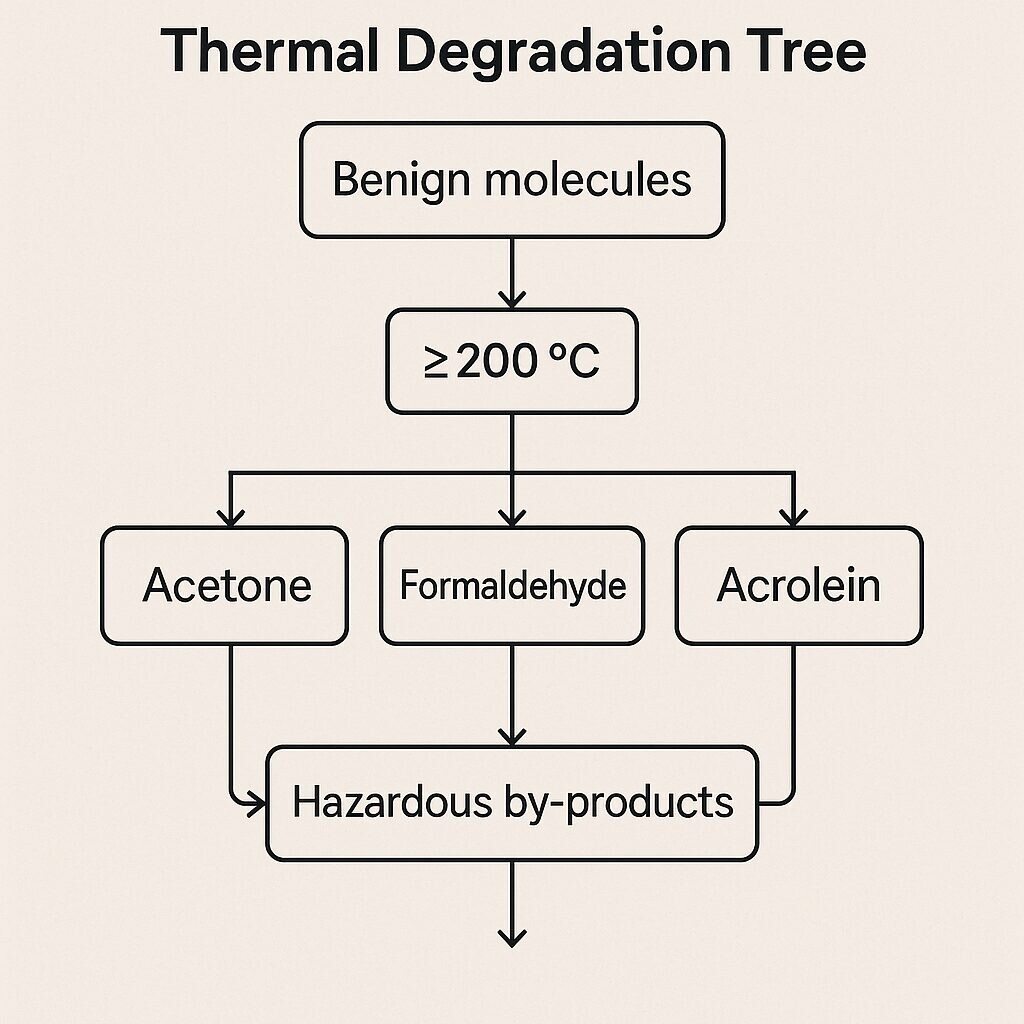
High-heat devices can degrade flavor molecules intotoxic or unknown byproducts, such as:
WhileGRAS (Generally Recognized as Safe)status is a baseline, it isnot enough. E-liquid flavorings should be:
Stay clear of:
Fewer components = lower risk. For PMTA in particular:
✅Recommended Option: “CUIGUAI Flavoringoffers pre-formulated, PMTA-ready flavorings that exclude restricted compounds while maintaining optimal sensory impact.”
Regulators demandevidence-based safety, not manufacturer claims. Make sure your flavoring meets the following:
Flavorings must withstand:
This iterative and evidence-driven workflow ensuresflavors are not only delightful but legally durable.
Compliance will soon be proactive. With the rise of AI in flavor formulation andin silico toxicology, manufacturers can:
Advanced systems likedigital twins of flavor matricesmay revolutionize regulatory submissions by linkinglab data to real-time compliance models.
As regulations become more comprehensive, designing e-liquid flavors is no longer a matter of creativity alone—it’s a complex balance oftoxicological caution, regulatory precision, and sensory science.
CUIGUAI Flavoringsupports manufacturers with a curated portfolio ofregulatory-compliant, pre-tested flavoringsdesigned for TPD, PMTA, and emerging standards. Build safer, smarter, and globally accepted flavors—without sacrificing taste.
Author: R&D Team, CUIGUAI Flavoring
Herausgegeben von: Guangdong Unique Flavor Co., Ltd.
Last Updated: May 29, 2025
Der Geschäftsumfang umfasst lizenzierte Projekte: Herstellung von Lebensmittelzusatzstoffen. Allgemeine Projekte: Verkauf von Lebensmittelzusatzstoffen; Herstellung von chemischen Produkten des täglichen Bedarfs; Verkauf von täglichen chemischen Produkten; technische Dienstleistungen, Technologieentwicklung, technische Beratung, Technologieaustausch, Technologietransfer und Technologieförderung; Forschung und Entwicklung von biologischen Futtermitteln; Forschung und Entwicklung zur industriellen Enzympräparation; Kosmetikgroßhandel; inländische Handelsagentur; Verkauf von Hygieneartikeln und medizinischen Einwegartikeln; Einzelhandel mit Küchenartikeln, Sanitärartikeln und Artikeln des täglichen Bedarfs; Verkauf von Artikeln des täglichen Bedarfs; Lebensmittelverkauf (nur Verkauf von vorverpackten Lebensmitteln).
Urheberrecht ©Guangdong Unique Flavour Co., Ltd.Alle Rechte vorbehalten. Datenschutzrichtlinie