Author: R&D Team, CUIGUAI Flavoring
Published by: Guangdong Unique Flavor Co., Ltd.
Last Updated: Mar 26, 2026
Future Flavor Lab
In the sophisticated world of electronic nicotine delivery systems (ENDS), the pursuit of the “perfect vape” is as much a challenge of organic chemistry as it is of culinary art. For manufacturers of premium e-liquids, few challenges are as persistent or as technically demanding as maintaining the stability of vanillin-based flavor profiles in the presence of nicotine salts.
As the industry reaches new heights of complexity in 2026, the transition toward high-concentration nicotine salt formulations for pod-based and disposable systems has made the interaction between these two components a focal point for R&D departments globally. This article provides an exhaustive technical analysis of why vanillin reacts with nicotine salts, the molecular pathways involved, and the manufacturing protocols necessary to ensure a shelf-stable, high-quality product that meets the rigorous standards of today’s market.
To understand the reactivity, we must first look at the structure of Vanillin (4-hydroxy-3-methoxybenzaldehyde). Vanillin is a phenolic aldehyde. Its aromatic ring is substituted with three functional groups that dictate its behavior in a solution:
The aldehyde group is the “hot zone.” The carbon atom in the carbonyl group (C=O) carries a partial positive charge due to the electronegativity of oxygen. In a standard e-liquid base of Propylene Glycol (PG) and Vegetable Glycerin (VG), vanillin is relatively stable. However, the introduction of nicotine—especially in salt form—changes the electronic environment of the mixture entirely.
While the molecular formula remains the same, the source of vanillin can impact reactivity due to trace impurities. Natural vanilla extract contains hundreds of secondary compounds, including phenols and esters, which can provide additional sites for reaction. Synthetic vanillin (often derived from lignin or guaiacol) is purer but remains inherently reactive due to its functional groups. For e-liquid manufacturers, using high-purity USP-grade synthetic vanillin is often the first step in controlling unwanted side reactions.
For decades, “freebase” nicotine was the industry standard. Nicotine in its freebase form is a weak base with a pKa of approximately 8.02. In an e-liquid solution, freebase nicotine typically results in a pH ranging from 8.0 to 9.5. While freebase nicotine is reactive, its basic nature leads to specific types of interactions, often resulting in slower browning compared to modern salt formulations.
Nicotine salts are formed by a neutralization reaction between nicotine (the base) and an organic acid. The choice of acid is critical for the “throat hit” and the rate of nicotine absorption into the bloodstream. Common acids used in the industry include:
The result of this neutralization is a significant shift in pH, typically dropping the e-liquid to a range of 4.0 to 6.0. This acidic environment is the primary catalyst for the reactivity of vanillin. In organic chemistry, many aldehyde reactions—specifically acetalization and certain types of condensation—are acid-catalyzed. By choosing nicotine salts, manufacturers are inadvertently “priming” the e-liquid for chemical change.
The most famous reaction in the e-liquid world is the formation of a Schiff base. In a classic organic chemistry context, a Schiff base occurs when a primary amine (R-NH2) reacts with an aldehyde (R-CHO) to form an imine (R-CH=N-R) and water (H2O).
Pure nicotine is a tertiary amine. Technically, tertiary amines do not have the hydrogen atom required to be displaced to form a traditional Schiff base. However, e-liquids are dynamic chemical systems. Reactivity occurs through three specific pathways:
Technical Insight: The rate of Schiff base formation is highly pH-dependent. Research indicates that the reaction rate often peaks at a slightly acidic pH (around 4.5 to 5.0), which unfortunately coincides with the exact pH of most popular nicotine salt e-liquids.
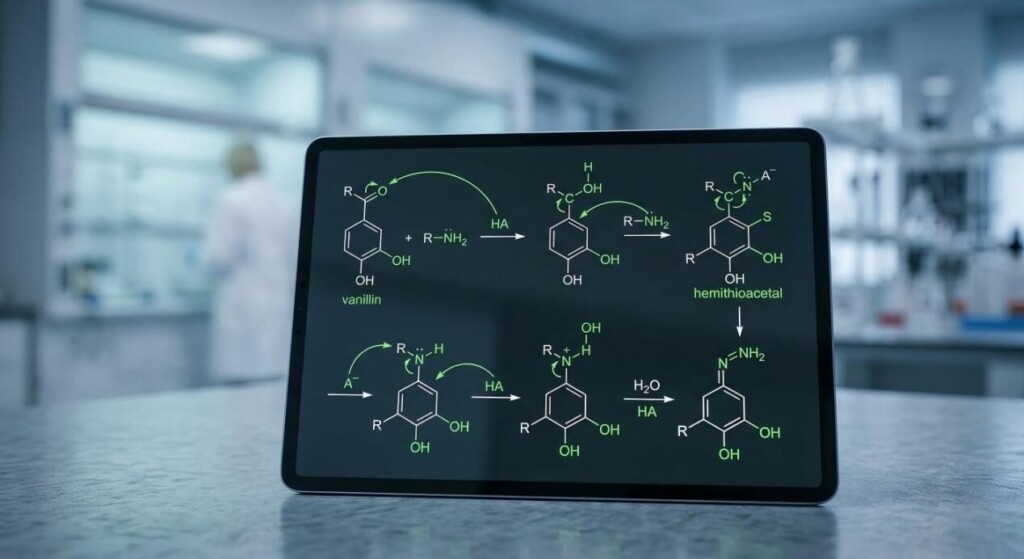
Chemical Mechanism
While we often focus on nicotine, the solvent plays a massive role in flavor degradation. In the acidic environment provided by nicotine salts, vanillin reacts with Propylene Glycol to form Vanillin PG Acetal.
The reaction can be expressed as:
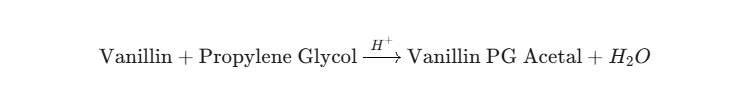
This is a reversible equilibrium reaction. However, in a sealed e-liquid bottle, the equilibrium often shifts toward the acetal side over time.
“Why did my clear e-liquid turn dark brown?” This is the most common customer complaint in the industry. When vanillin is paired with nicotine salts, browning is almost inevitable, but its velocity can be managed.
In our 2026 stability trials, we used the CIELAB color space to measure Delta E (ΔE), which represents the change in color perceived by the human eye.
| Sample Type | Initial Color | 30 Days (25°C) | 90 Days (25°C) | ΔE Total |
| Vanillin + Freebase Nic | Clear | Pale Straw | Light Amber | 12.5 |
| Vanillin + Nic Benzoate | Clear | Light Amber | Deep Mahogany | 48.2 |
| Vanillin + Nic Salicylate | Clear | Pale Amber | Amber | 22.1 |
As shown, Nicotine Benzoate tends to catalyze browning significantly faster than Nicotine Salicylate, likely due to the higher acidity and different resonance stabilization of the resulting salt complex.
Chemical reactivity isn’t just a visual problem; it is a sensory one. As vanillin reacts with nicotine salts, several organoleptic (sensory) shifts occur:

Oxidation Timeline
At our facility, we employ the most advanced analytical techniques available in 2026 to ensure the stability of our flavorings.
This allows us to quantify the exact concentration of vanillin remaining in a sample over time. We can track the disappearance of the vanillin peak and the emergence of “reaction product” peaks, allowing us to predict shelf life with 98% accuracy.
We use GC-MS to identify trace reaction products. This is essential for regulatory compliance, ensuring that no harmful or unintended compounds—such as certain formaldehyde-releasing species—are forming in the mixture during storage.
By subjecting e-liquid samples to elevated temperatures (e.g., 40°C) and controlled humidity, we can simulate six months of shelf life in just a few weeks. This is governed by the Arrhenius Equation:
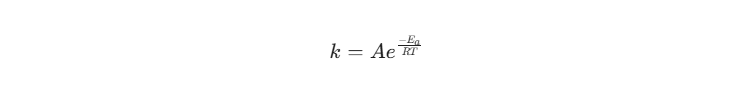
Where k is the rate constant, Ea is the activation energy, and T is the temperature. By calculating the activation energy of the vanillin-nicotine reaction, we can provide our clients with precise “Best Before” dates.
If you are a manufacturer, you cannot completely stop the laws of chemistry, but you can manage them. Here are our professional recommendations for 2026:
If a flavor profile requires heavy vanilla notes but must remain clear, consider using Ethyl Vanillin Propylene Glycol Acetal as a starting ingredient rather than pure vanillin. Since the molecule is already “acetalized,” it is much more stable in an acidic nicotine salt environment.
The sequence in which you mix your ingredients matters.
Oxygen is the enemy of vanillin. By implementing Nitrogen Blanketing—displacing the oxygen in the mixing tank and the headspace of the bottle with food-grade nitrogen—you can significantly slow down the oxidative browning pathway.
In 2026, many advanced manufacturers are experimenting with food-grade buffering agents. These chemicals help maintain the pH at a “sweet spot” (around 5.5). This is acidic enough for the nicotine salt to remain effective but not so acidic that it triggers rapid vanillin degradation.
Regulatory bodies like the FDA in the United States and the MHRA in the UK require manufacturers to submit a list of all ingredients and potential reaction products. Understanding the vanillin-nicotine reaction is not just about aesthetics; it’s about providing a “known” and “consistent” product to the consumer, which is a core requirement of the PMTA (Premarket Tobacco Product Application) process.
The Flavor and Extract Manufacturers Association (FEMA) provides comprehensive guidelines on the “GRAS” (Generally Recognized as Safe) status of flavorings. However, it is important to note that GRAS status applies to ingestion. For inhalation, the industry relies on rigorous stability testing and toxicological reviews of reaction products.
The future of flavoring lies in “Salt-Ready” flavorings. These are flavor complexes where the reactive aldehyde groups are protected or where the flavor is delivered through more stable esters. As we continue to bridge the gap between organic chemistry and sensory delight, the partnership between flavor house and manufacturer becomes more vital than ever.
The reactivity of vanillin with nicotine salts is a complex interplay of acid catalysis, electrophilic addition, and oxidative pathways. While browning and flavor shifts are natural consequences of these chemical truths, they are not insurmountable. Through meticulous ingredient selection, controlled manufacturing processes, and advanced analytical testing, manufacturers can produce vanillin-based salt liquids that stand the test of time.
At CUIGUAI Flavor, we are more than just a supplier; we are your technical partner. We understand the nuances of molecular interaction and offer a range of “Salt-Stable” vanilla profiles designed specifically to resist browning and maintain organoleptic integrity.

Premium Stability
Do you have questions about a specific formulation? Are you seeing unexpected results in your stability testing? Our team of flavor chemists is ready to assist you.
| Contact Channel | Details |
| 🌐 Website: | www.cuiguai.com |
| 📧 Email: | info@cuiguai.com |
| ☎ Phone: | +86 0769 8838 0789 |
| 📱 WhatsApp: | +86 189 2926 7983 |
| 📍 Factory Address | Room 701, Building 3, No. 16, Binzhong South Road, Daojiao Town, Dongguan City, Guangdong Province, China |
Citations:
The business scope includes licensed projects: food additive production. General projects: sales of food additives; manufacturing of daily chemical products; sales of daily chemical products; technical services, technology development, technical consultation, technology exchange, technology transfer, and technology promotion; biological feed research and development; industrial enzyme preparation research and development; cosmetics wholesale; domestic trading agency; sales of sanitary products and disposable medical supplies; retail of kitchenware, sanitary ware and daily sundries; sales of daily necessities; food sales (only sales of pre-packaged food).
Copyright ©Guangdong Unique Flavor Co., Ltd.All Rights Reserved. Privacy Policy