Author: R&D Team, CUIGUAI Flavoring
Published by: Guangdong Unique Flavor Co., Ltd.
Last Updated: Feb 07, 2026
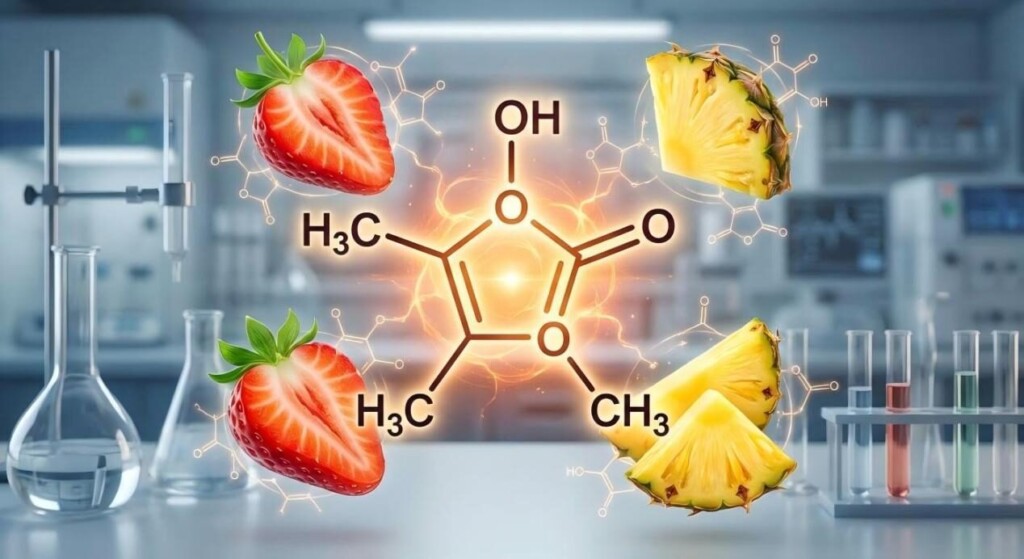
The Furaneol Molecule Secret
In the intricate world of flavor chemistry, where molecular structures dictate sensory experiences, certain compounds stand out for their profound impact. Among these, furaneol, specifically 2,5-dimethyl-4-hydroxy-3(2H)-furanone, often abbreviated as DMHF or simply furaneol, holds a paramount position, particularly in the realm of fruit flavors. While sugars provide the foundational sweetness, it is furaneol that imbues fruits like strawberries and pineapples with their characteristic, rich, and intensely sweet aromatic notes, elevating their flavor profile beyond mere saccharine taste. For manufacturers of specialized e-liquid flavorings, understanding the nuanced role of furaneol is not just beneficial, but essential for crafting authentic, complex, and highly desirable fruit profiles. This comprehensive exploration delves into the chemical properties of furaneol, its biosynthesis in fruits, its sensory attributes, synergistic interactions with other flavor compounds, and its indispensable application in flavor formulation, particularly within the e-liquid industry.
Furaneol is a heterocyclic organic compound, a derivative of furan, characterized by its distinctive furanone ring structure. Its full chemical nomenclature, 2,5-dimethyl-4-hydroxy-3(2H)-furanone, precisely describes its arrangement of carbon, hydrogen, and oxygen atoms, which ultimately dictates its unique sensory properties.
The core of furaneol’s identity lies in its 3(2H)-furanone ring. The presence of two methyl groups at positions 2 and 5, and a hydroxyl group at position 4, are critical for its characteristic aroma. It’s important to note that furaneol exists in two tautomeric forms: the dominant keto form and a less prevalent enol form. While both forms contribute to its overall profile, the keto form is generally considered more stable and is primarily responsible for its sensory impact. The specific stereochemistry of the molecule, particularly the orientation of the methyl groups, can also influence subtle differences in its perceived aroma, although these are often less significant than the fundamental presence of the molecule itself.
At room temperature, pure furaneol is a white to off-white crystalline solid. It possesses a relatively low melting point (around 77-80°C) and is highly soluble in water and organic solvents such as ethanol and propylene glycol, making it exceptionally versatile for flavor formulation. Its volatility is moderate, allowing it to contribute significantly to both initial aroma perception and lingering aftertaste.
Chemically, furaneol is quite reactive due to its furanone ring and hydroxyl group. It can undergo various reactions, including oxidation and degradation, particularly under conditions of high heat or prolonged exposure to light and oxygen. This chemical instability is a critical consideration for flavor manufacturers, as it necessitates careful handling, storage, and formulation strategies to preserve its integrity and desired sensory contribution in e-liquid applications. Stabilization techniques, such as encapsulation or the use of antioxidants, are often employed to extend its shelf life and maintain its flavor profile.
The presence of furaneol in fruits is not accidental; it is a meticulously orchestrated biochemical process. While its exact biosynthetic pathway can vary slightly between fruit species, a common precursor is carbohydrate metabolism, particularly from hexoses like glucose and fructose.
One of the most well-established pathways for furaneol synthesis involves the Maillard reaction, a complex series of non-enzymatic browning reactions between amino acids and reducing sugars. While the Maillard reaction is more commonly associated with cooked foods, certain enzymatic reactions in fruits can mimic aspects of this process or contribute to the formation of precursors.
More specifically, in strawberries, the biosynthesis of furaneol is thought to involve the degradation of certain sugars and amino acids, with intermediates like 4-hydroxy-5-methyl-3(2H)-furanone (norfuraneol) serving as precursors. Norfuraneol can then be methylated to form furaneol. This enzymatic methylation step is crucial and highlights the intricate biochemical machinery at play within the fruit. Research has also indicated that certain plant enzymes, such as 4-hydroxy-2,5-dimethyl-3(2H)-furanone synthase, are directly involved in its production. The activity of these enzymes is influenced by various factors, including fruit ripeness, growing conditions, and even genetic predisposition.
In pineapples, while the exact pathway shares similarities, there can be subtle differences in the precursor availability and enzymatic activity, leading to varying concentrations and contributions of furaneol to the overall flavor profile. The pineapple plant (Ananas comosus) accumulates significant amounts of furaneol as it ripens, contributing to its characteristic sweet and tropical notes.
Several factors can significantly influence the concentration of furaneol in fruits, directly impacting their perceived sweetness and aroma:
Understanding these biosynthetic pathways and influencing factors is paramount for flavor chemists. By mimicking or enhancing these natural processes, or by carefully selecting and processing raw materials, manufacturers can optimize the furaneol content in their flavorings, ensuring a more authentic and potent product.
Furaneol is a powerhouse of flavor, renowned for its highly desirable sensory attributes that are central to the appeal of many fruits.
The primary descriptor for furaneol’s aroma is caramel-like, strawberry-like, fruity, and sweet. It possesses a warm, cooked sugar note reminiscent of butterscotch or burnt sugar, coupled with an intensely jammy and ripe fruit character. This complex profile is what makes it so indispensable. It doesn’t just add sweetness; it adds a depth and richness of sweetness that simple sugars cannot replicate.
While primarily an aroma compound detected orthonasally (through the nose) and retronasally (through the back of the mouth to the nose), furaneol also influences taste perception. Its intense sweetness at low concentrations can directly enhance the perception of sweetness on the tongue, even beyond the direct stimulation of taste receptors by sugars. This phenomenon, known as cross-modal enhancement, means that the brain interprets the presence of furaneol’s sweet aroma as an amplification of the actual sweet taste. This makes it an invaluable tool for flavorists seeking to create perceptions of intense sweetness without necessarily increasing sugar content.
Furaneol has a remarkably low odor detection threshold, meaning even minuscule concentrations can be perceived by the human nose. Its reported odor threshold in air is around 0.005 ppb (parts per billion), and in water, it can be as low as 0.04 ppm (parts per million). This high potency is a significant advantage for flavor formulators, as it allows for impactful flavor delivery with relatively small amounts of the compound, contributing to cost-effectiveness and avoiding unwanted off-notes that might arise from higher concentrations of less potent ingredients.
Precision Flavor Formulation
Flavors are rarely the product of a single compound. Instead, they arise from complex interactions between numerous volatile and non-volatile molecules. Furaneol is a master collaborator, exhibiting significant synergistic effects with other flavor compounds, particularly in strawberry and pineapple.
Furaneol acts as a crucial “sweet anchor” in strawberry flavor, providing a foundation upon which other volatile compounds build.
The magic happens when these compounds are present in the right ratios. A strawberry flavor rich in esters but lacking furaneol might taste vibrant but thin, lacking the characteristic jammy sweetness. Conversely, too much furaneol without sufficient supporting esters could lead to a one-dimensional, overly cooked-sugar profile.
Similarly, in pineapple, furaneol works in harmony with a diverse array of compounds to create its iconic tropical sweetness.
The synergistic interplay is particularly evident in the transition from fresh pineapple to processed forms like canned pineapple or pineapple juice. Heat processing can significantly increase furaneol concentration through Maillard-like reactions, which is why canned pineapple often has a more pronounced caramelized sweetness compared to fresh fruit. This highlights the importance of temperature and processing in modulating furaneol levels and, consequently, the perceived flavor.
Beyond direct aroma contribution, the synergistic effect of furaneol on perceived sweetness is a powerful tool for flavorists. By enhancing the perception of sweetness without adding caloric sugars, furaneol becomes a vital component in developing reduced-sugar or sugar-free e-liquid formulations that still deliver an impactful sweet fruit experience. This is particularly relevant in an industry where consumers are increasingly health-conscious but unwilling to compromise on flavor intensity.
For manufacturers of specialized e-liquid flavorings, the precise application of furaneol is a cornerstone of crafting premium, authentic fruit profiles. Its versatility and potency make it an indispensable ingredient.
The primary objective for any e-liquid flavor manufacturer is to replicate the natural taste experience of fruits. In strawberries and pineapples, this is almost impossible without furaneol.
As previously discussed, furaneol significantly enhances the perception of sweetness. This allows flavorists to formulate e-liquids that taste intensely sweet without relying solely on added sweeteners, which can sometimes introduce off-notes or have regulatory implications. This is particularly valuable for creating “sweetened” fruit profiles that still remain clean and true to the fruit.
Beyond sweetness, furaneol also contributes to the overall mouthfeel of an e-liquid. Its warm, slightly syrupy notes can impart a sense of richness and fullness, making the vapor feel more substantial and satisfying. This “body” is crucial for a complete sensory experience.
When incorporating furaneol into e-liquid formulations, several technical considerations are paramount:

Visualizing Flavor Synergy
While strawberries and pineapples are prime examples of fruits where furaneol plays a starring role, its influence extends far wider across the natural world and into various food and beverage applications. Its presence contributes to the characteristic sweetness and aroma of:
This widespread occurrence underscores furaneol’s fundamental importance as a natural flavor compound. Its ability to impart desirable sweet and caramel notes makes it a valuable asset not just for fruit flavors, but across a vast spectrum of culinary applications. For flavor manufacturers, this breadth of application means that a deep understanding of furaneol’s chemistry and sensory impact is a truly cross-functional asset.
The scientific community continues to unravel the complexities of furaneol. Ongoing research focuses on several key areas:
For specialized e-liquid flavor manufacturers, tracking these advancements is paramount. New discoveries in furaneol research can lead to innovative formulation strategies, more potent and stable flavorings, and the ability to meet evolving consumer demands for increasingly authentic and complex taste experiences. This ongoing scientific exploration is what drives the cutting edge of flavor creation.
At CUIGUAI Flavor, we recognize that creating exceptional e-liquid flavorings requires a profound understanding of individual flavor compounds and their intricate interactions. Our team of expert flavor chemists possesses extensive knowledge and experience in working with furaneol, leveraging its unique properties to craft unparalleled strawberry and pineapple (and many other fruit) flavor profiles.
We meticulously source high-quality furaneol and integrate it into our formulations with precision, ensuring optimal concentration and synergistic balance with other key aroma chemicals. Our commitment to rigorous quality control, sensory evaluation, and adherence to the latest scientific advancements means that our clients receive flavorings that are not only authentic and potent but also consistent and stable.
We understand that the e-liquid market demands innovation and excellence. By mastering the art and science of furaneol application, we provide our partners with a competitive edge, enabling them to deliver e-liquids that captivate consumers with their true-to-fruit sweetness and rich aromatic complexity. Whether you seek the luscious jamminess of a ripe strawberry or the vibrant, caramelized sweetness of a tropical pineapple, our expertise in furaneol ensures your flavorings will stand out.
Furaneol, 2,5-dimethyl-4-hydroxy-3(2H)-furanone, is far more than just another flavor compound; it is a critical determinant of the rich, sweet, and authentic aroma and taste profiles of fruits like strawberries and pineapples. Its unique chemical structure, intricate biosynthesis, potent sensory attributes, and remarkable synergistic interactions with other volatiles make it an indispensable tool for flavor chemists.
For the specialized e-liquid flavoring industry, understanding and expertly utilizing furaneol is crucial for crafting products that genuinely resonate with consumers. Its ability to provide deep, jammy sweetness, enhance perceived sweetness, and contribute to mouthfeel ensures that e-liquids deliver an unparalleled and authentic fruit vaping experience. As flavor science continues to evolve, furaneol will undoubtedly remain a cornerstone ingredient, continually refined and optimized to meet the ever-growing demand for sophisticated and delightful sensory journeys. Its unsung role is, in fact, the very secret to the success of countless beloved fruit flavors, both in nature and in expertly crafted applications.

Premium Product Presentation
Ready to elevate your e-liquid flavors with authentic, complex fruit profiles? Contact our expert flavor scientists today to discuss your specific needs, request technical assistance, or receive free samples of our premium flavorings. Discover how our mastery of compounds like furaneol can transform your products.
| Contact Channel | Details |
| 🌐 Website: | www.cuiguai.com |
| 📧 Email: | info@cuiguai.com |
| ☎ Phone: | +86 0769 8838 0789 |
| 📱 WhatsApp: | +86 189 2926 7983 |
| 📍 Factory Address | Room 701, Building 3, No. 16, Binzhong South Road, Daojiao Town, Dongguan City, Guangdong Province, China |
The business scope includes licensed projects: food additive production. General projects: sales of food additives; manufacturing of daily chemical products; sales of daily chemical products; technical services, technology development, technical consultation, technology exchange, technology transfer, and technology promotion; biological feed research and development; industrial enzyme preparation research and development; cosmetics wholesale; domestic trading agency; sales of sanitary products and disposable medical supplies; retail of kitchenware, sanitary ware and daily sundries; sales of daily necessities; food sales (only sales of pre-packaged food).
Copyright ©Guangdong Unique Flavor Co., Ltd.All Rights Reserved. Privacy Policy