Author: R&D Team, CUIGUAI Flavoring
Published by: Guangdong Unique Flavor Co., Ltd.
Last Updated: Mar 03, 2026

Coil Activation
In the sophisticated world of electronic liquid (e-liquid) manufacturing, we are often preoccupied with the delicate balance of top, heart, and base notes. We discuss “mouthfeel,” “throat hit,” and “aromatic longevity.” However, there is a darker, more adhesive reality lurking beneath the surface of every tank and pod: the “gunk.” For years, both hobbyist vapers and industrial manufacturers have casually blamed “caramelization” for the blackened, crusty residue that inevitably claims the life of a heating coil.
But as we peel back the layers of organic chemistry and thermal dynamics, a more complex culprit emerges. The question isn’t just about heat; it’s about a specific chemical transformation that defines the culinary world—from the golden crust of a sourdough loaf to the savory sear on a Wagyu steak. We are talking about the Maillard reaction.
As a premier manufacturer of fragrances for the vaping industry, we believe it is vital to move beyond layman’s terms. Does this sophisticated dance of sugars and amino acids actually occur within the microscopic environment of a vaping coil? And if it does, what does that mean for the fragrances we meticulously craft? This deep dive explores the intersections of organic chemistry, thermodynamics, and sensory science to answer once and for all: Is the Maillard reaction the true “coil killer”?
To diagnose what is happening on a 0.15-ohm mesh coil at 60 watts, we must first establish a rigorous chemical baseline. In common parlance, any heat-induced browning is called “caramelization.” In chemistry, this is a significant oversimplification.
Caramelization is the pyrolysis of sugar. It is a strictly “solo” performance by carbohydrates. When you heat sucrose, glucose, or fructose to high temperatures—typically above 160℃ (320℉)—the molecules break down, release water (dehydration), and reform into complex, high-molecular-weight polymers like caramelans (C12H18O9), caramelens (C36H50O25), and caramelins (C125H188O80). This provides that classic, sweet-bitter “burnt sugar” note and a deep brown color.
The Maillard reaction, however, is a “duet.” It requires two lead actors: a reducing sugar (like glucose or fructose) and an amino group (typically from an amino acid, protein, or certain alkaloids). Named after the French chemist Louis-Camille Maillard in 1912, this reaction is far more chemically active at lower temperatures than caramelization and produces a significantly wider and more complex array of flavor and aroma compounds.
The Maillard reaction is not a single event but a cascade of reactions:
A common rebuttal to the Maillard theory in vaping is: “E-liquids are made of Propylene Glycol (PG), Vegetable Glycerin (VG), and flavorings. There are no proteins or amino acids. Therefore, it can’t be Maillard.”
While logical at first glance, this overlooks the chemical reality of Nicotine and the hidden complexities of Fragrance Chemistry.
Nicotine (C10H14N2) is a tertiary amine. While it is not a primary amino acid, it is a nitrogen-rich alkaloid. In the high-energy environment of a heating coil, nicotine does not remain inert. Thermal energy can cause nicotine to act as a catalyst or even a participant in browning reactions. Research has shown that nitrogenous compounds, even those that aren’t traditional proteins, can catalyze the degradation of sugars and interact with the carbonyl groups found in various flavoring components.
As a manufacturer, we know that “Natural Strawberry Flavor” is rarely just one molecule. It is often a botanical extract containing trace amounts of organic matter, including microscopic amounts of proteins or free amino acids. Even at parts-per-million (ppm) levels, these nitrogenous “impurities” are more than sufficient to trigger Maillard-style browning when subjected to repeated heating cycles.
Propylene Glycol and Vegetable Glycerin are alcohols (polyols). Under extreme heat, especially in the presence of oxygen and metal catalysts (the coil wire), they can oxidize into aldehydes and ketones—such as formaldehyde, acetaldehyde, and acrolein. These “new” carbonyl groups are highly reactive and look for any nitrogen source to begin the Maillard sequence
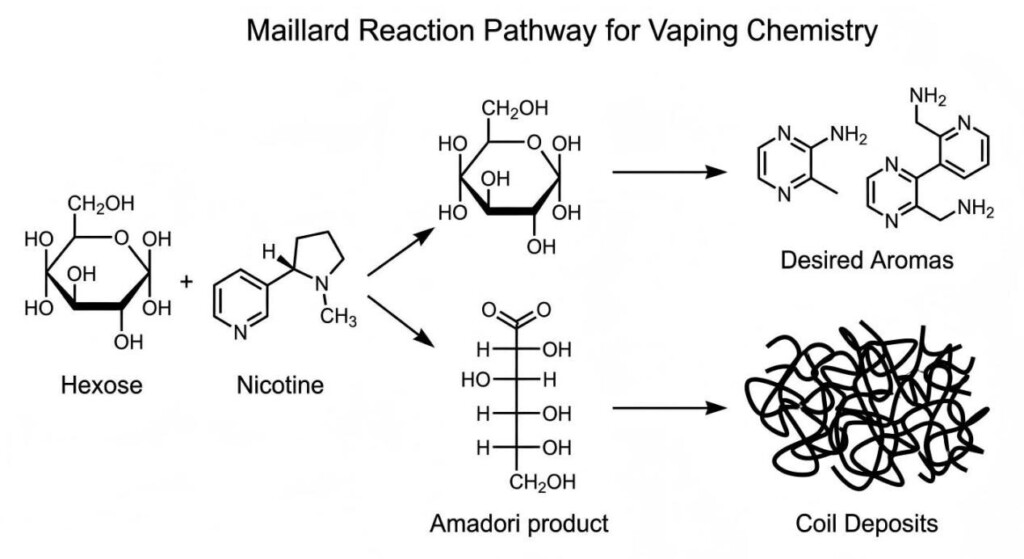
Maillard Diagram
The modern vaping device is not just a heater; it is a micro-scale chemical reactor. To understand why Maillard happens, we must look at the thermodynamics of the coil-wick interface.
When a vaper presses the fire button, the coil reaches temperatures far exceeding the boiling point of the e-liquid. However, the liquid does not evaporate uniformly. Due to the Leidenfrost effect, a thin, insulating layer of vapor forms between the red-hot metal and the liquid. This allows the coil to reach temperatures of 200℃ to 300℃ almost instantaneously.
The liquid droplets that manage to bridge this vapor gap and touch the metal are subjected to “flash” heating. This provides more than enough activation energy for the Maillard reaction to proceed at lightning speed—seconds instead of the hours usually required in an oven.
The materials used in coils—Kanthal (Iron-Chromium-Aluminum), Nichrome (Nickel-Chromium), and Stainless Steel—are not just resistors; they are catalysts. Transition metals like Nickel and Iron are known to lower the activation energy for oxidation and polymerization reactions. This means that the metal wire itself is “helping” the e-liquid turn into gunk.
As the e-liquid in the wick vaporizes, it leaves behind the heavier, less volatile components. Over a few hundred puffs, the concentration of sugars, nicotine, and flavorings in the wick increases significantly. This “enriched” liquid becomes a thick, syrupy concentrate that is even more prone to the Maillard reaction.
“Significant amounts of toxic metals… leak from some e-cigarette heating coils and are present in the aerosols… fresher coils give off metals more readily, but the chemical complexity of the residual liquid increases significantly over time.” — Study: Lead and Other Toxic Metals Found in E-Cigarette ‘Vapors’ | Johns Hopkins
If amino groups are the spark, sweeteners are the fuel. Most “coil killer” liquids share one common trait: a high concentration of Sucralose.
Sucralose (C12H19Cl3O8) is a chlorinated disaccharide. While it is stable at room temperature, it is notoriously unstable when heated. When sucralose reaches approximately 120℃ (248℉), it begins to decompose, releasing hydrogen chloride (HCl) gas.
This HCl acts as an acid catalyst, which drastically accelerates both the caramelization of any other sugars present and the Maillard reaction between flavor aldehydes and nicotine. The result is a rapid transition from a clear liquid to a dark, tar-like substance on the coil.
Other common additives like Ethyl Maltol (which provides a “cotton candy” or “jammy” sweetness) are also reactive. While not as destructive as sucralose, they contribute to the overall “carbon load” on the coil. As these molecules break down, they form the “building blocks” of the blackened crust.
Research published in PMC (National Center for Biotechnology Information) indicates that sucralose-enhanced degradation is a primary driver of aerosol toxicity. The presence of sucralose not only increases the production of aldehydes but also creates an acidic environment that facilitates the polymerization of flavor molecules into the solid deposits we call gunk.
The Maillard reaction is famous for creating “savory,” “toasted,” and “nutty” flavors. In the culinary world, this is a miracle (think of roasted coffee). In the vaping world, it is a sensory disaster.
The Maillard reaction produces a class of compounds called pyrazines. In controlled amounts, pyrazines are used in tobacco-flavored e-liquids to give them a “dry” or “toasted” finish. However, when they are produced uncontrollably on a coil through the degradation of a “Strawberry Cream” or “Blueberry Muffin” liquid, they clash horribly with the intended flavor profile.
This is why, as a coil ages, the flavor loses its “brightness” and begins to taste “earthy,” “burnt,” or “ashy.” You are literally tasting the byproduct of the Maillard reaction.
Often, vapers believe they have “vaper’s tongue” (olfactory fatigue). While this exists, many cases are actually the result of the e-liquid’s flavor profile shifting mid-tank. The delicate esters and aldehydes of the fragrance have been replaced by the heavy, dominant pyrazines and furans of the Maillard reaction.

Coil Comparison
At our fragrance manufacturing facility, we don’t just “make things smell good.” We engineer for the reality of the heating coil. Understanding that the Maillard reaction does happen allows us to take proactive steps to prevent it.
Many popular flavor molecules are aldehydes (like Cinnamaldehyde for cinnamon or Vanillin for vanilla). Aldehydes are highly reactive and are a primary component in the Maillard sequence. To combat this, we often utilize acetals. Acetals are “masked” versions of aldehydes that are much more stable in the bottle and in the tank. They only release the flavor when aerosolized, keeping the coil clean during the liquid phase.
We employ advanced molecular distillation and cold-filtration techniques for our natural extracts. By removing trace proteins and nitrogenous compounds at the source, we effectively “starve” the Maillard reaction of its amino partners.
We guide our clients away from high-percentage sucralose. Instead, we offer proprietary “sweetness enhancers” that rely on olfactory triggers rather than physical sugars. If a sweetener must be used, we recommend those with higher thermal stability thresholds and lower reactivity with nicotine.
Since the Maillard reaction is pH-dependent (it thrives in alkaline environments), we carefully balance the acidity of our fragrance concentrates. By keeping the e-liquid slightly on the acidic side, we can slow down the Amadori rearrangement and extend coil life significantly.
“The content, purity, identities and doses of flavour molecules… are not clear. Typically, flavourings promote the production of free radicals… individual ENDS flavourants induce toxicity.” — Emerging ENDS products and challenges in tobacco control toxicity research – PMC
As we look toward 2026 and beyond, the industry is moving toward a “total system” approach.
Does the Maillard reaction happen on the coil? The answer is a definitive and scientifically backed “Yes.” While simple caramelization plays a role, it is the Maillard reaction—fueled by nicotine, flavor aldehydes, and trace impurities—that is responsible for the complex, foul-tasting, and adhesive “gunk” that plagues the vaping experience. It is a testament to the fact that vaping is not just a mechanical act, but a complex chemical process.
For the vaper, this means choosing liquids with high-quality, stable fragrances. For the e-liquid brand owner, it means partnering with a fragrance manufacturer that understands the molecular science of the coil. By acknowledging the Maillard reaction, we can move toward a future of cleaner, safer, and more consistent flavor.

R&D Laboratory
Are you a manufacturer looking to solve the “coil gunk” problem? Do you want to develop a premium line of e-liquids that maintains its flavor profile from the first puff to the last? Our technical team is ready to provide the chemical expertise you need.
| Contact Channel | Details |
| 🌐 Website: | www.cuiguai.com |
| 📧 Email: | info@cuiguai.com |
| ☎ Phone: | +86 0769 8838 0789 |
| 📱 WhatsApp: | +86 189 2926 7983 |
| 📍 Factory Address | Room 701, Building 3, No. 16, Binzhong South Road, Daojiao Town, Dongguan City, Guangdong Province, China |
The business scope includes licensed projects: food additive production. General projects: sales of food additives; manufacturing of daily chemical products; sales of daily chemical products; technical services, technology development, technical consultation, technology exchange, technology transfer, and technology promotion; biological feed research and development; industrial enzyme preparation research and development; cosmetics wholesale; domestic trading agency; sales of sanitary products and disposable medical supplies; retail of kitchenware, sanitary ware and daily sundries; sales of daily necessities; food sales (only sales of pre-packaged food).
Copyright ©Guangdong Unique Flavor Co., Ltd.All Rights Reserved. Privacy Policy