Author: R&D Team, CUIGUAI Flavoring
Published by: Guangdong Unique Flavor Co., Ltd.
Last Updated: Mar 12, 2026

Scientist at GC-MS
In the competitive landscape of e-liquid manufacturing, quality isn’t just a regulatory checkbox; it’s the foundation of brand reputation and consumer trust. While sensory evaluation—the “taste test”—remains crucial, the ultimate authority on what constitutes a flavoring’s true profile and purity lies within the analytical data. This data is delivered via Gas Chromatography (GC), often coupled with Mass Spectrometry (MS).
Understanding how to read a GC report is an indispensable skill for any e-liquid manufacturer seeking consistency, safety, and innovation. This technically-rich guide will demystify the GC/MS process, walk you through the anatomy of a profiling report, and provide actionable insights into leveraging this data to ensure uncompromised flavor quality.
For years, the flavor industry operated behind a veil of proprietary blends. In the e-liquid sector, this lack of transparency is rapidly becoming a relic. Manufacturers must know exactly what they are putting into their products, not just for compliance with emerging regulations (like the FDA’s PMTA process in the US or the TPD in Europe), but to ensure that Batch B tastes identical to Batch A, and that no undesirable compounds are present.
Gas Chromatography is the gold standard for this level of analysis. It provides a molecular fingerprint, allowing us to see the individual components that create complex flavor profiles. This guide moves beyond the basic definitions and dives deep into how you can use this analytical tool as a powerful quality control and R&D asset.
Before we dissect the report, it is vital to understand the technology that generates it. Gas Chromatography-Mass Spectrometry (GC-MS) is a two-step analytical method used to separate and identify individual chemical substances within a complex sample.
The process begins with the chromatograph. The flavor sample is injected into a heated injector port, where it is instantly vaporized. A carrier gas (usually Helium or Hydrogen), known as the mobile phase, sweeps the vaporized sample into a column.
The column is a long, narrow tube coated internally with a substance known as the stationary phase. The various chemical compounds in the flavor sample have different affinities for the stationary phase. As the mobile phase moves the sample through the column, different compounds interact with the stationary phase to varying degrees.
This variation in travel speed achieves the physical separation of the complex mixture into its individual constituents.
As the separated compounds emerge individually from the GC column, they enter the Mass Spectrometer. This is the “identification” engine.
Within the MS, the molecules are bombarded with a beam of electrons, causing them to break apart into charged fragments (ions). This process is known as ionization. These fragments are then accelerated and sorted based on their mass-to-charge ratio (m/z) using electromagnetic fields.
The detector records the relative abundance of each fragment, producing a “mass spectrum.” Every chemical compound produces a unique, reproducible fragmentation pattern—a molecular “fingerprint.” The MS software then compares this spectrum against vast electronic libraries (such as the NIST library) to provide a definitive identification of the compound.
When you receive a GC report from a lab, you are typically presented with two main sections: the visual chromatogram and the data table (often called the Peak Table).
The chromatogram is the graphic representation of the separation process.
In a complex e-liquid flavoring, the chromatogram will feature numerous peaks, ranging from large, dominating peaks (major components like PG or main flavor notes) to tiny, almost imperceptible “baseline” peaks (minor or trace components).
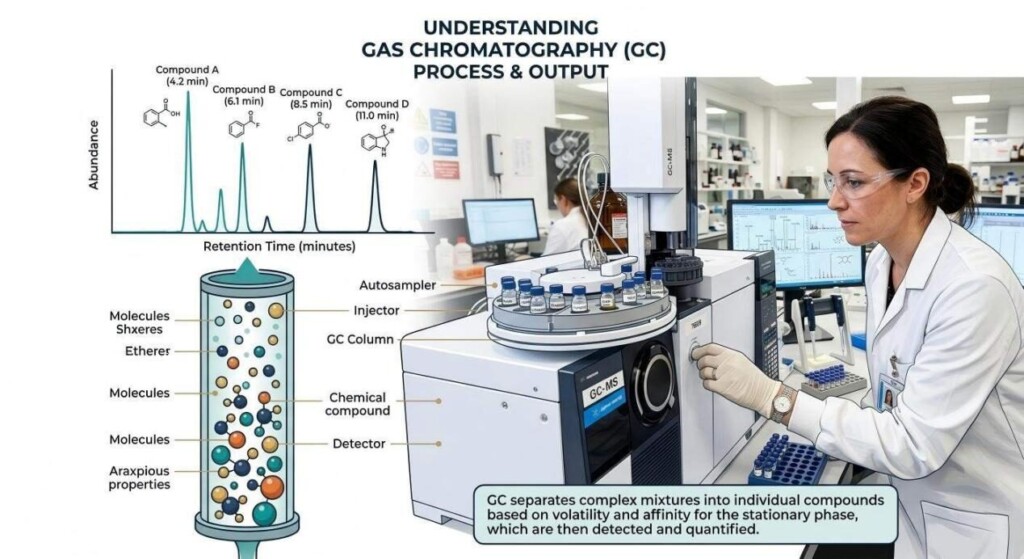
GC Infographic
While Retention Time is essential for a specific lab running a specific method, it is not universally reproducible. Small differences in column length, flow rate, or temperature programming can shift RTs.
To standardize this, chemists use the Retention Index (RI). The RI (often called the Kovats Index) normalizes retention times relative to the elution of a standard series of n-alkanes analyzed under the same conditions. This makes RI a much more robust and transferable value for identifying compounds across different labs and systems.
According to research published on websites of academic institutions like Wikipedia’s entry on the Kovats Retention Index, RI values are stable across different gas chromatographic systems as long as the stationary phase of the column remains the same, providing a critical metric for quality assurance.
The data table accompanying the chromatogram is where the quantitative and precise qualitative data resides. As an e-liquid manufacturer, this is the data you must master.
The lab arbitrarily assigns a number to each detected peak, usually in chronological order of elution.
This column lists the name of the chemical compound identified by the Mass Spectrometer and confirmed by library searching. For flavoring agents, these names will be specific aromatic molecules (e.g., Isoamyl Acetate for banana, Ethyl Butyrate for pineapple).
The CAS registry number is a unique numerical identifier for a chemical substance. It is the gold standard for specificity, eliminating confusion caused by different chemical synonyms. For example, “Ethyl 3-methylbutyrate” and “Ethyl isovalerate” are the same compound; the CAS number 108-64-5 provides a single, unambiguous reference.
This column represents the integrated area under the peak on the chromatogram. The area is proportional to the concentration of the compound. Laboratories use this value to calculate the relative percentage of each component.
This is a critical quantitative metric. It shows the percentage of the total detected signal that is attributable to a specific compound. It is calculated as:
(Area of Specific Peak / Total Area of All Peaks) * 100
While % Area does not give you an absolute concentration (like mg/mL), it is an excellent metric for relative quantification. It answers the question: “Of the total flavoring, how much of it is this specific ester?”
** persistive Note:** For batch-to-batch consistency, comparing the % Area of key flavor components is paramount. If your signature “Strawberry Ripple” flavoring relies on 15% Ethyl Methylphenylglycidate, and a new batch shows 10%, your sensory profile will be different.
When the Mass Spectrometer software compares the unknown sample’s mass spectrum to the library reference, it calculates a matching score or Quality Factor, often expressed on a scale of 0 to 100 or 0 to 1000.
Always look for high match factors on key aromatic compounds.
Often, reports include both the Experimental RI (the one calculated from your sample) and the Library/Reference RI. Comparing these two values provides a second layer of confirmation for identification, supplementing the MS match data.
Now that we understand the report’s structure, how do we use this information to ensure the quality of e-liquid flavorings?
Consistency is the benchmark of a professional e-liquid manufacturer. GC reports are your tool for ensuring your suppliers are delivering a consistent product.
The Strategy: Establish a “Gold Standard” profile. When you find a flavoring batch that is perfect, archive its GC report. For every new shipment of that flavoring, require a new GC report and compare the main peaks’ Area % to your Gold Standard. Major deviations (often >10% relative difference in key peaks) should be flagged and discussed with the supplier.
While flavorings are complex, premium products must be clean. GC reports allow you to screen for undesirable compounds. This includes:
The most critical safety screening in the e-liquid industry is for diketones, specifically Diacetyl (2,3-Butanedione) and Acetyl Propionyl (2,3-Pentanedione, or AP). These compounds, associated with “popcorn lung” (bronchiolitis obliterans) when inhaled, are frequently found in buttery or creamy flavor profiles.
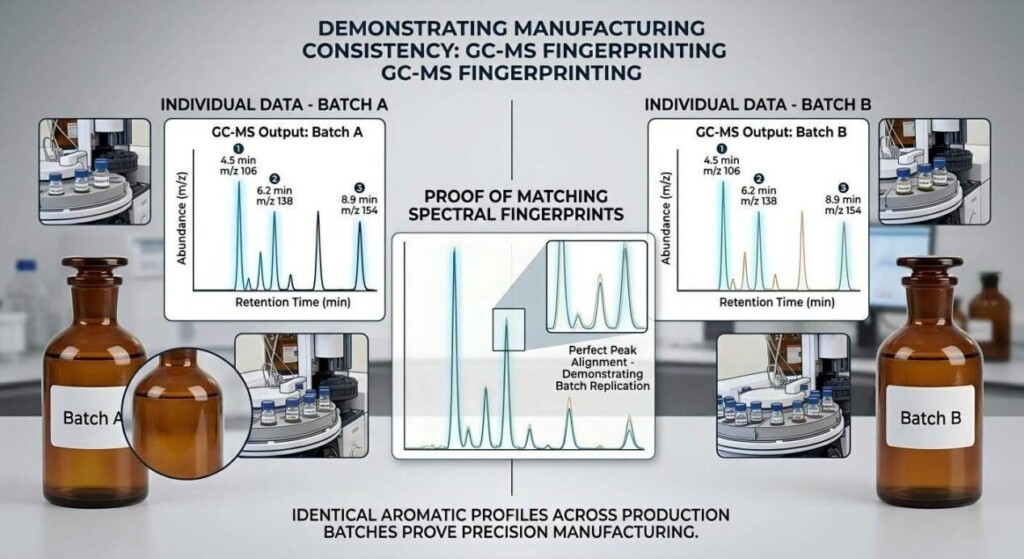
Batch Consistency
Standard GC-MS, while capable of detecting these molecules, often requires specialized sample preparation (like derivatization) or specific detector settings (like Electron Capture Detection, or ECD) to achieve the necessary low detection limits (often in the low parts-per-million, ppm range) for strict safety compliance.
While general GC profiling reports provide an excellent overview of the flavor composition, they are not always optimized as “safety certificates” for low-level diketone detection.
Beyond consistency and purity, GC reports are an excellent educational tool for flavorists. By studying the GC profiles of complex flavorings, you can begin to deconstruct why a certain “custard” tastes richer or a specific “mint” has a sharper cooling effect. You can see the relationship between chemical structures and sensory experience.
For example, observing a high concentration of vanillin (CAS 121-33-5) and ethyl vanillin (CAS 121-32-4) in a vanilla custard profile explains its sweetness and depth, but finding acetoin (CAS 513-86-0) provides the buttery texture.
To illustrate the power of GC reading, let’s consider a hypothetical analysis of a complex e-liquid flavoring: “Spiced Apple Fritter.”
While GC-MS is incredibly powerful, it is not a standalone magic wand for flavor quality. It is a complementary tool that must work alongside sensory analysis.
Mastering the skill of reading GC reports empowers you to demand more from your flavor suppliers. You are no longer relying on simple assurances; you are demanding data-driven proof of quality.
A premier flavoring manufacturer doesn’t just provide flavorings; they provide transparency. They understand that their GC profile is their contract of quality with you. They utilize robust, validated analytical methods, they maintain rigorous in-house quality standards that leverage GC data, and they are willing to provide detailed COAs (Certificates of Analysis) and GC profiles upon request for their core products. This transparency is a key indicator of their confidence in their own manufacturing processes and the chemical integrity of their flavorings.
By demanding and scrutinizing this data, you elevate your own production, ensure the safety and consistency of your e-liquids, and ultimately build a stronger, more trustworthy brand in a mature market. For comprehensive industry guidelines on safety and quality management, organizations such as the International Fragrance Association (IFRA) offer resources that also apply to flavor manufacturing principles.

R&D Collaboration
We invite e-liquid manufacturers to engage in technical exchange. Requesting our latest GC profiles for your signature flavors is the first step toward superior consistency. We offer free sample kits tailored to your specific profile needs.
Contact Us for Superior Flavor Integrity:
| Contact Channel | Details |
| 🌐 Website: | www.cuiguai.com |
| 📧 Email: | info@cuiguai.com |
| ☎ Phone: | +86 0769 8838 0789 |
| 📱 WhatsApp: | +86 189 2926 7983 |
| 📍 Factory Address | Room 701, Building 3, No. 16, Binzhong South Road, Daojiao Town, Dongguan City, Guangdong Province, China |
The business scope includes licensed projects: food additive production. General projects: sales of food additives; manufacturing of daily chemical products; sales of daily chemical products; technical services, technology development, technical consultation, technology exchange, technology transfer, and technology promotion; biological feed research and development; industrial enzyme preparation research and development; cosmetics wholesale; domestic trading agency; sales of sanitary products and disposable medical supplies; retail of kitchenware, sanitary ware and daily sundries; sales of daily necessities; food sales (only sales of pre-packaged food).
Copyright ©Guangdong Unique Flavor Co., Ltd.All Rights Reserved. Privacy Policy