Author: R&D Team, CUIGUAI Flavoring
Published by: Guangdong Unique Flavor Co., Ltd.
Last Updated: Mar 23, 2026

Analytical Laboratory
In the rapidly evolving landscape of the 2026 e-liquid and inhalation formulation industry, manufacturers have moved far beyond the elementary “fruit and menthol” pairings of the past decade. As consumer palates mature, demanding highly complex, multi-layered organoleptic experiences, the chemical complexity of the flavor concentrates themselves has skyrocketed. Simultaneously, regulatory scrutiny from international health bodies and the U.S. Food and Drug Administration (FDA) has intensified, specifically regarding the stability, safety, and physical behavior of aerosolized compounds under thermal stress.
For modern flavor manufacturers and e-liquid formulators, the ultimate technical challenge lies in managing the delicate, often volatile equilibrium between hydrophilic (water-attracting/polar) and hydrophobic (water-repelling/non-polar) compounds.
Achieving this critical balance is not merely a matter of subjective taste; it is a fundamental prerequisite for physical stability, predictable aerosolization performance, chemical safety, and regulatory compliance. A poorly balanced formulation inevitably leads to phase separation, muted or distorted flavor profiles, oxidative degradation, and the accelerated degradation of heating elements (coils). In this definitive guide, we will dissect the fundamental chemistry, thermodynamic principles, solubilization strategies, and manufacturing protocols necessary to master the hydrophilic-hydrophobic balance in commercial e-liquid production.
To fully comprehend the mechanics of flavor balance, we must first deeply analyze the thermodynamic and chemical environment in which these flavor molecules reside: the base matrix. In almost all commercial applications, this matrix is a binary solvent system composed of Propylene Glycol (PG) and Vegetable Glycerin (VG).
Propylene Glycol (IUPAC name: propane-1,2-diol; chemical formula C3H8O2) is an aliphatic, synthetic organic compound that belongs to the diol family. The presence of two hydroxyl (-OH) groups makes PG a highly hydrophilic and polar molecule. It is miscible with water, alcohols, and many organic solvents.
Because PG has a relatively low molecular weight (76.09 g/mol) and lower dynamic viscosity compared to VG, it allows for exceptionally rapid molecular diffusion. In the terminology of flavor chemistry, PG is the optimal “flavor carrier.” Its polarity enables it to form strong hydrogen bonds with a vast array of polar flavor molecules, such as naturally derived acids, simple esters, and alcohols. When formulated correctly, PG ensures that these hydrophilic compounds remain in a stable, homogeneous, and thermodynamically favorable solution, preventing premature crystallization or precipitation.
Vegetable Glycerin (IUPAC name: propane-1,2,3-triol; chemical formula C3H8O3), often simply referred to as glycerol, is a naturally occurring polyol compound possessing three hydroxyl groups. While VG is technically completely miscible with water and PG, its unique molecular structure creates a distinctly different solubility environment for flavor compounds.
VG is highly viscous, dense, and possesses a highly interlinked network of internal hydrogen bonding. While excellent for producing dense vapor clouds due to its humectant properties and thermal behavior, VG is fundamentally less effective at solvating non-polar, hydrophobic flavor compounds. In formulations that rely heavily on VG (such as the prevalent 70/30 or 80/20 VG/PG ratios favored for Sub-Ohm devices), manufacturers frequently encounter the phenomenon of “flavor fallout.”
Flavor fallout occurs when hydrophobic aromatic compounds—unable to form sufficient intermolecular bonds with the polyol matrix—begin to self-associate and aggregate. Over time, these aggregates form microscopic droplets, breaking the emulsion and leading to a cloudy appearance, or worse, distinct “oily” phases floating at the air-liquid interface of the bottle.
To predict how a flavor molecule will behave in a PG/VG matrix, chemists rely on the Octanol-Water Partition Coefficient, commonly expressed as LogP.
The partition coefficient is defined mathematically as the ratio of concentrations of a compound in a mixture of two immiscible solvents at equilibrium. By standard convention, these solvents are 1-octanol (a non-polar, lipophilic solvent) and water (a polar, hydrophilic solvent).
The formula is expressed as:
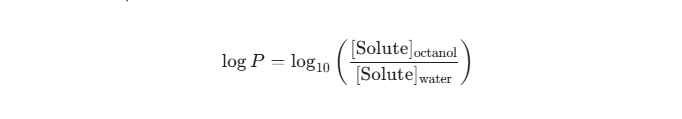
Understanding the LogP of your individual flavor isolates is the first step in predictive formulation, moving the process from trial-and-error to applied chemistry.
Hydrophilic compounds are the high-note heroes of an e-liquid profile. They provide the immediate, sharp, and vibrant flavor bursts that consumers perceive upon inhalation. Because they actively seek out hydrogen bonds, they integrate seamlessly into the PG phase of the carrier matrix.
These compounds are highly polar due to their carboxylic acid groups.
Molecular Flavor Interactions
Hydrophobic molecules represent the bold, complex, and lingering base notes of an e-liquid. In recent years, as the industry has shifted heavily toward authentic, botanically derived flavors, the use of highly lipophilic compounds has surged. These non-polar molecules naturally repel the polar PG/VG carrier, seeking instead to bond with other non-polar molecules.
Terpenes are highly volatile, unsaturated hydrocarbons found widely in the essential oils of plants.
How does a master formulator keep a high-LogP citrus oil seamlessly integrated into a high-VG, heavily polar base without phase separation? The solution lies in chemical “bridges”—co-solvents that feature both hydrophilic and lipophilic properties.
Triacetin is an indispensable tool in the modern flavoring toolkit. Chemically, it is the triester of glycerol and acetic acid. It possesses a unique amphiphilic-like quality, allowing it to act as a mediating agent.
High-purity, food-grade ethanol (Ethyl Alcohol) is a highly effective, albeit controversial, co-solvent.
For consumers with sensitivities to Propylene Glycol, the industry has turned to 1,3-Propanediol. While it functions similarly to PG in its solvent capabilities, its slightly altered carbon structure gives it a slightly different solubility profile, sometimes requiring adjustments in the hydrophilic/hydrophobic flavor ratios to maintain the exact same organoleptic profile as a PG-based liquid.
The formulation of a perfectly balanced e-liquid is not a static achievement; it is a dynamic equilibrium that is constantly threatened by environmental factors.
As commercial e-liquids are manufactured, warehoused, and shipped globally, they encounter massive temperature fluctuations. “Winterization” is a severe threat to e-liquid stability.
Thermodynamically, the solubility of hydrophobic molecules in a polar solvent decreases as the temperature drops. If a formulator has created a liquid that is “on the edge” of its maximum hydrophobic load at room temperature (22℃), exposing that liquid to a cold night in a delivery truck (4℃) will lower the kinetic energy of the system.
This drop in energy causes nucleation. The hydrophobic flavor molecules or heavily saturated hydrophilic sweeteners (like Sucralose or Ethyl Maltol) will literally “crash out” of the solution, crystallizing or forming cloudy agglomerations. Once crashed out, simple shaking at room temperature is rarely sufficient to redissolve them completely; thermal energy (heating the liquid) combined with mechanical agitation is required to reverse the process.
Even if an emulsion appears stable immediately after mixing, microscopic droplets of hydrophobic oils may still exist within the matrix. Over time, due to a phenomenon known as Ostwald Ripening, smaller droplets will thermodynamically dissolve and redeposit onto larger droplets to minimize the total surface area and surface energy. Eventually, this coalescence leads to macro-scale phase separation—the dreaded “layer of oil” at the top of an old bottle of e-liquid.

E-Liquid Production Process
When the hydrophilic/hydrophobic balance fails and phase separation occurs, the formulation faces a much more insidious threat than just poor taste: rapid chemical degradation.
Hydrophobic oils (particularly terpenes and aldehydes) have a lower specific gravity than the PG/VG carrier matrix. Therefore, when they separate, they migrate upwards to the air-liquid interface—the headspace of the bottle.
This surface exposure is disastrous. The flavor oils are now in direct, concentrated contact with atmospheric oxygen trapped in the bottle.
Terpenes like Limonene are highly susceptible to autoxidation. When exposed to oxygen and ambient light, Limonene degrades into various oxides and carvone derivatives. Organoleptically, this transforms a bright, fresh, zesty lemon flavor into a harsh, chemical note that consumers frequently compare to “furniture polish” or “floor cleaner.”
A perfectly balanced e-liquid traps these delicate terpene molecules securely within the dense, oxygen-resistant network of the PG/VG matrix, shielding them from the headspace air and vastly extending the product’s shelf life.
The end-user cares very little for LogP values, thermodynamic instability, or triacetin ratios. They care entirely about the sensory result. The hydrophilic/hydrophobic balance dictates every facet of the vaping experience.
As we navigate the highly regulated landscape of 2026, regulatory bodies have adopted zero-tolerance policies for ambiguous formulation data. The FDA’s Center for Tobacco Products (CTP) and overarching human food safety programs have refined their requirements for Premarket Tobacco Product Applications (PMTA).
According to current FDA regulatory frameworks, e-liquid manufacturers can no longer rely on opaque, “proprietary blend” safety sheets from flavor houses. There is a mandate for absolute molecular transparency.
Regulatory submissions now require comprehensive data proving that a specific flavor formulation remains stable over its entire stated shelf life. This means manufacturers must utilize advanced analytical chemistry to prove their hydrophilic/hydrophobic balance is maintained.
If a manufacturer submits a PMTA for a product that demonstrates phase separation during an accelerated 6-month stability test, that product will be summarily rejected based on the unpredictable toxicological profile of vaping separated flavor oils.
Knowing the chemistry is only half the battle; executing it on an industrial scale requires rigorous Standard Operating Procedures. Simple magnetic stirring is entirely inadequate for commercial e-liquid production in 2026.
Never dump all ingredients into a master batch simultaneously. Always isolate your most stubborn, high-LogP hydrophobic compounds and dissolve them into your pure Propylene Glycol (and any required co-solvents like Triacetin) first. This creates a highly concentrated “flavor base.” Only once this base is optically crystal clear and verified homogeneous should it be introduced to the heavier Vegetable Glycerin phase.
To forcefully integrate the lighter PG-flavor base into the dense VG base, mechanical force is required. High-shear homogenizers operate at massive RPMs (typically 10,000 to 30,000 RPM). The rotor blades force the liquid through a stationary stator screen, subjecting the fluid to immense hydraulic shear and cavitation. This physically tears the hydrophobic oil droplets apart, reducing their particle size from the macro-scale (visible) down to the sub-micron level, creating a kinetically stable microemulsion.
For ultra-premium lines, passing the homogenized liquid through an inline ultrasonic flow cell utilizes high-frequency sound waves to further reduce particle size to the nano-scale. Nanoemulsions are incredibly stable and drastically improve flavor transfer and aerosolization efficiency.
Implement a strict formulation limit. Formulators should calculate the total percentage of high-LogP compounds in any given recipe. If the “hydrophobic load” exceeds 15-20% of the total flavor concentrate volume in a Max VG blend, the recipe should be automatically flagged for co-solvent adjustment or reformulating to prevent inevitable fallout.
To illustrate the real-world application of these principles, consider a recent challenge faced by a mid-sized e-liquid brand attempting to launch a “Lemon Basil Gelato” profile in an 80/20 VG/PG base.
Our formulation experts intervened with a three-step chemical rescue:
Q: Can I use distilled water to balance my hydrophilic and hydrophobic compounds?
A: Distilled water is the ultimate polar solvent. While adding 1-3% distilled water to a high-VG mix can dramatically lower viscosity and aid in wicking, it actually worsens the hydrophobic separation problem. Water will fiercely repel lipid-based or heavy terpene compounds. It should be used for viscosity control, not as a flavor co-solvent.
Q: How do I know if my flavor concentrate is separating in the master batch tank?
A: Visually, look for a “lensing” effect—small, clear circular lenses floating on the surface of the bulk liquid. You may also notice the liquid looks “milky” or opalescent when light is shone through it, a classic sign of macro-emulsion failure. Analytically, taking samples from the top, middle, and bottom of the tank and running them through an HPLC will quickly reveal if the heavy flavor molecules are floating to the top.
Q: Does steeping affect the hydrophilic/hydrophobic balance?
A: “Steeping” is essentially allowing time for the chemical reactions (like esterification between alcohols and acids) to reach thermodynamic equilibrium, and for off-gassing of highly volatile unwanted top-notes (like ethyl alcohol used in the extraction process). Proper steeping does not “fix” a broken emulsion; if a liquid is separated, steeping will only allow it to separate further. Proper mechanical homogenization is required before steeping begins.
The quest for the perfect commercial e-liquid is, at its core, a quest for molecular harmony. As the industry pushes toward more complex, authentic, and naturally derived flavor profiles, the fundamental conflict between water-loving and water-repelling compounds will only intensify.
By deeply understanding the partition coefficients of your raw materials, intelligently deploying co-solvents like Triacetin, and investing in high-shear homogenization equipment, formulators can force these opposing chemical forces into a stable, lasting alliance.
As we navigate the stringent regulatory and competitive landscape of 2026, the manufacturers who invest in the rigorous chemistry behind the clouds will be the ones who define the future of the inhalation flavor industry. Excellence is no longer achieved by accident; it is engineered molecule by molecule.

Aether Essence Harmony Blend
At CUIGUAI Flavor, we don’t just supply flavors; we supply the chemical expertise required to make them work flawlessly in your specific matrix. Whether you are struggling with flavor muting in Max-VG blends, looking to stabilize a complex botanical profile, or need analytical assurance for your 2026 PMTA submissions, our team of Ph.D. chemists and master flavorists is ready to assist.
| Contact Channel | Details |
| 🌐 Website: | www.cuiguai.com |
| 📧 Email: | info@cuiguai.com |
| ☎ Phone: | +86 0769 8838 0789 |
| 📱 WhatsApp: | +86 189 2926 7983 |
| 📍 Factory Address | Room 701, Building 3, No. 16, Binzhong South Road, Daojiao Town, Dongguan City, Guangdong Province, China |
The business scope includes licensed projects: food additive production. General projects: sales of food additives; manufacturing of daily chemical products; sales of daily chemical products; technical services, technology development, technical consultation, technology exchange, technology transfer, and technology promotion; biological feed research and development; industrial enzyme preparation research and development; cosmetics wholesale; domestic trading agency; sales of sanitary products and disposable medical supplies; retail of kitchenware, sanitary ware and daily sundries; sales of daily necessities; food sales (only sales of pre-packaged food).
Copyright ©Guangdong Unique Flavor Co., Ltd.All Rights Reserved. Privacy Policy