Author: R&D Team, CUIGUAI Flavoring
Published by: Guangdong Unique Flavor Co., Ltd.
Last Updated: Mar 07, 2026

Lab Research
The electronic cigarette industry has undergone profound changes over the past decade, driven by rapid technological innovation, shifting consumer preferences, and evolving regulatory landscapes. However, no single event has had a more significant and lasting impact on the industry’s trajectory than the emergence of Vitamin E Acetate as a contaminant and the subsequent public health crisis.
For manufacturers of electronic liquids (e-liquids) and their upstream suppliers, such as fragrance and flavoring specialists, understanding the full context of Vitamin E Acetate is not merely a matter of historical recall. It is a fundamental requirement for operating with integrity, ensuring product safety, and maintaining the trust of both regulators and consumers. This detailed blog post explores the history of Vitamin E Acetate, the pivotal ban that followed the EVALI crisis, and the robust, technologically-driven response from the legitimate vaping industry.
To understand the crisis, it is first necessary to understand the substance at its center. Vitamin E Acetate, chemically known as alpha-tocopheryl acetate, is a synthetic form of Vitamin E. It is an ester created from alpha-tocopherol (the active form of Vitamin E) and acetic acid.
In its intended applications, Vitamin E Acetate is a ubiquitous and generally safe compound. It is highly valued for its antioxidant properties and stability.
For decades, Vitamin E Acetate has been a staple ingredient in the cosmetic and skincare industries. It is found in countless lotions, creams, serums, and sunscreens, where it is used to moisturize the skin and protect it from free radical damage. In these topical applications, its safety profile is well-established.
Furthermore, Vitamin E Acetate is widely used as a dietary supplement, often found in multivitamin capsules. When ingested orally, the body breaks down the acetate ester, releasing alpha-tocopherol, which is essential for numerous physiological functions.
However, the safety of a substance is inherently linked to its route of administration. This is the crucial distinction that was overlooked, with tragic consequences.
(Citation Source: the Cosmetic Ingredient Review or a pharmaceutical journal article on tocopheryl acetate.)
Vitamin E Acetate was never an intended or approved ingredient in standard nicotine-containing e-liquids produced by reputable manufacturers. The constituents of quality e-liquids are well-known: propylene glycol (PG), vegetable glycerin (VG), high-purity nicotine, and food-grade flavoring agents.
The problem arose in the unregulated, illicit market, primarily involving cannabis (THC) vaping cartridges.
Starting around 2018 and escalating in 2019, illicit manufacturers of THC cartridges sought a way to dilute (or “cut”) their expensive THC oil without compromising the product’s appearance. THC oil is naturally thick and viscous. Traditional cutting agents like PG or VG are much thinner, and their presence in significant quantities would make the resulting product visibly runny, a clear sign to consumers of dilution and poor quality.
Illicit operators discovered that Vitamin E Acetate possessed physical properties—specifically its high viscosity and clear appearance—that made it a perfect deceptive diluent. It mimicked the thickness of pure THC oil, allowing unscrupulous manufacturers to significantly cut their products with a cheap additive while maintaining the illusion of high potency.
This misuse was driven purely by profit, with no consideration for the physiological implications of inhaling vaporized Vitamin E Acetate. The chemical that was safe for skin application and ingestion was about to be introduced into human lungs.
The catastrophic consequences of this deceptive practice began to manifest in mid-2019. Health departments across the United States started reporting clusters of severe, unexplained respiratory illnesses associated with vaping. This outbreak came to be known as EVALI: E-cigarette or Vaping Use-Associated Lung Injury.
The symptoms were severe and rapidly progressing, including shortness of breath, cough, chest pain, fever, fatigue, and, in some cases, gastrointestinal issues. Patients often presented with significantly reduced oxygen saturation and required hospitalization, frequently necessitating supplemental oxygen or mechanical ventilation.
The EVALI crisis triggered an intense, multi-agency investigation led by the Centers for Disease Control and Prevention (CDC) and the Food and Drug Administration (FDA), in collaboration with state and local health departments.
The initial investigation was challenging because patients were often reluctant to disclose their use of illicit THC products. However, as data accrued, a powerful pattern emerged. The vast majority of EVALI patients reported using vaping products, and a significant preponderance (often 80% or more in various data subsets) reported using products containing THC.
The breakthrough came when public health laboratories began analyzing product samples obtained from patients and, crucially, the bronchoalveolar lavage (BAL) fluid—fluid samples collected from the lungs of hospitalized patients.
Analysis of BAL fluid provided definitive evidence. Studies conducted by the CDC and academic partners consistently found Vitamin E Acetate in the lung fluid of EVALI patients. For example, a key study published in the New England Journal of Medicine analyzed BAL fluid from 51 EVALI patients and found Vitamin E Acetate in 48 (94%) of them. It was not found in the BAL fluid of healthy control groups.
(Citation Source: Blount BC, et al. Vitamin E Acetate in Bronchoalveolar-Lavage Fluid Associated with EVALI. N Engl J Med. 2020.)
The findings were clear. Vitamin E Acetate was the primary, though perhaps not exclusive, chemical of concern driving the EVALI outbreak.
Why is inhaling Vitamin E Acetate so dangerous? While research is ongoing, the scientific community has identified several key mechanisms:
The combination of these physical and chemical impacts on the delicate lung environment explained the severe injuries observed in EVALI patients.
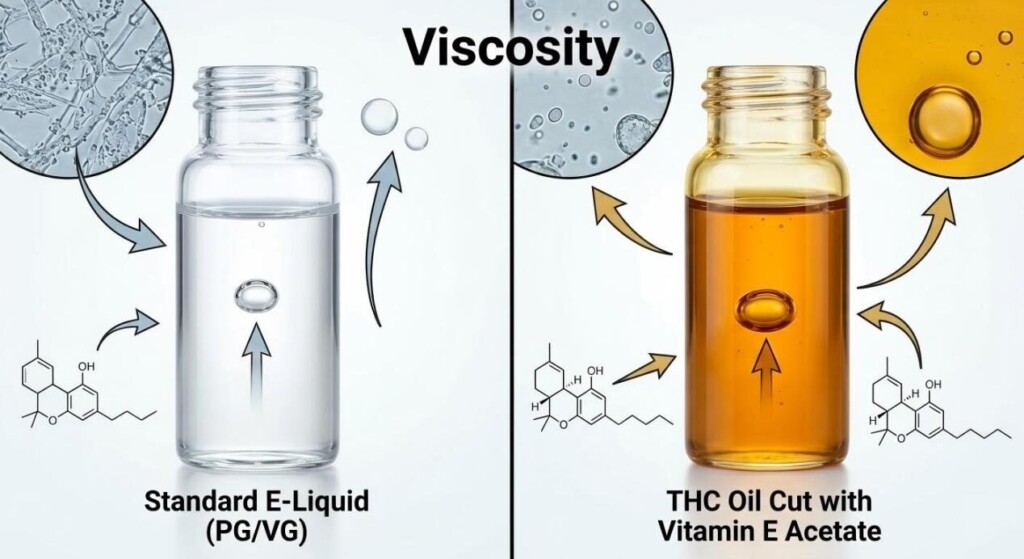
Viscosity Guide
The definitive link between Vitamin E Acetate and EVALI spurred immediate and aggressive regulatory action. The primary goal was to remove Vitamin E Acetate-containing products from the market and to educate the public about the extreme dangers of illicit vaping products, especially those containing THC.
In the United States, several states implemented emergency bans on the sale of all flavored vaping products and, more specifically, any product containing unauthorized additives like Vitamin E Acetate.
The FDA issued strong warnings to consumers, advising them to stop using THC-containing vaping products and to avoid any vaping products bought “off the street” or from illicit sources. The agency also utilized its enforcement authority to crack down on the manufacture and distribution of unauthorized vaping products, particularly those implicated in the EVALI crisis.
(Citation Source: The official FDA “Lung Illnesses Associated with Use of Vaping Products” page.)
The EVALI crisis and the subsequent regulatory response were a defining moment. It highlighted the severe risks of an unregulated market and the critical importance of strict ingredient control.
The EVALI crisis was a seismic event for the entire vaping industry. While the primary culprit was the illicit THC market, the entire sector, including legitimate nicotine e-liquid manufacturers and flavor suppliers, was scrutinized.
The response from the professional, regulated industry was swift and decisive. It was an opportunity to demonstrate a commitment to consumer safety and to differentiate the legitimate market from the dangerous operators that caused the crisis.
Reputable manufacturers recognized that maintaining public trust required unprecedented levels of transparency regarding product composition. The mindset shifted from simply complying with existing rules to proactively exceeding them.
As fragrance and flavor experts providing the aromatic heart of e-liquids, our role became more critical than ever. We formalized and amplified our commitment to purity. We reaffirmed that Vitamin E Acetate has absolutely no place in any of our flavoring concentrates. Our focus has always been on complex, high-purity flavoring compounds derived from food-safe sources, and the EVALI crisis only reinforced the necessity of this strict approach.
The cornerstone of the industry’s response has been the widespread adoption of rigorous analytical testing. It was no longer enough to simply trust ingredient suppliers; manufacturers had to verify.
The primary tool in this verification process is Gas Chromatography-Mass Spectrometry (GC/MS). GC/MS is the gold standard for chemical analysis, capable of separating, identifying, and quantifying the individual components of complex mixtures.

GC/MS Analysis
In a GC/MS analysis, a sample of an e-liquid or flavor concentrate is first vaporized and passed through a long column (the Gas Chromatograph). Different compounds within the sample move through the column at different rates based on their physical properties, effectively separating them.
As the compounds emerge from the column, they enter the Mass Spectrometer. Here, they are bombarded with electrons, causing them to break apart into charged fragments (ions). The mass spectrometer measures the mass-to-charge ratio of these fragments, creating a unique “fingerprint” (a mass spectrum) for each compound.
By comparing these fingerprints to vast reference libraries, toxicologists and analytical chemists can definitively identify every compound present in a sample. Crucially, they can screen for specific substances of concern, such as:
GC/MS analysis provides a scientific guarantee of product purity. It is an indispensable component of any modern, safety-conscious e-liquid quality control program. Legitimate manufacturers and flavor suppliers incorporate GC/MS testing into multiple stages of the production process, from raw material validation to finished product testing.
Beyond testing, the EVALI crisis accelerated the development and adoption of standardized best practices within the industry.
Manufacturers, often in collaboration with industry trade associations, developed comprehensive quality management systems (QMS). These systems emphasize:
These industry-led initiatives demonstrate a proactive commitment to safety that goes beyond mere regulatory compliance.
(Citation Source: The Vapor Technology Association’s (VTA) guidance on quality control.)
The industry’s response has been a process of introspection, reform, and technological advancement, all aimed at one goal: ensuring that nothing like EVALI can happen again.
The story of Vitamin E Acetate and its role in the EVALI crisis is a powerful and sobering chapter in the history of the vaping industry. It is a cautionary tale of the dangers inherent in an unregulated market and the profound consequences of misusing a common chemical.
However, it is also a story of a robust, technologically sophisticated, and principled response. The legitimate vaping industry, including fragrance and flavoring manufacturers like ourselves, has emerged from this crisis stronger, more transparent, and more focused on consumer safety than ever before.
By understanding the history of Vitamin E Acetate, implementing stringent ingredient controls, and leveraging advanced analytical technologies like GC/MS, we can ensure that integrity and safety remain the foundation of our industry. As we look to the future, our commitment to innovation is unwavering, but it will always be guided by the paramount principle of protecting public health.

Clean Facility
Ensuring the absolute purity of e-liquids and their flavoring components is not just a regulatory requirement; it is a moral imperative. We are committed to leading the industry in flavoring safety and technological innovation.
We invite e-liquid manufacturers, regulators, and industry stakeholders to engage with us. Whether you seek technical exchange regarding advanced GC/MS testing protocols, wish to request free samples of our rigorously tested, high-purity flavor concentrates, or simply have questions about our quality management systems, we are here to collaborate.
Contact our team today to explore how we can work together to ensure the highest standards of safety and quality in your products.
Contact Us:
| Contact Channel | Details |
| 🌐 Website: | www.cuiguai.com |
| 📧 Email: | info@cuiguai.com |
| ☎ Phone: | +86 0769 8838 0789 |
| 📱 WhatsApp: | +86 189 2926 7983 |
| 📍 Factory Address | Room 701, Building 3, No. 16, Binzhong South Road, Daojiao Town, Dongguan City, Guangdong Province, China |
The business scope includes licensed projects: food additive production. General projects: sales of food additives; manufacturing of daily chemical products; sales of daily chemical products; technical services, technology development, technical consultation, technology exchange, technology transfer, and technology promotion; biological feed research and development; industrial enzyme preparation research and development; cosmetics wholesale; domestic trading agency; sales of sanitary products and disposable medical supplies; retail of kitchenware, sanitary ware and daily sundries; sales of daily necessities; food sales (only sales of pre-packaged food).
Copyright ©Guangdong Unique Flavor Co., Ltd.All Rights Reserved. Privacy Policy