Author: R&D Team, CUIGUAI Flavoring
Published by: Guangdong Unique Flavor Co., Ltd.
Last Updated: Mar 13, 2026

Chirality Header
In the high-precision world of e-liquid flavoring, the difference between a “premium” experience and a “medicinal” one often comes down to a few atoms pointing in the wrong direction. As a manufacturer committed to sensory excellence, we understand that the foundation of any great vape juice is not just the chemical formula, but the stereochemical architecture of the molecules involved.
Among the myriad of compounds used in the flavor industry, Menthol stands as a titan. It is the cooling agent of choice, the backbone of “ice” flavors, and a crucial component for providing the “throat hit” that former smokers often crave. However, many formulators are surprised to learn that “Menthol” is not a single entity. It is a family of isomers, and in the world of chirality, your “left hand” and your “right hand” are worlds apart.
This technical deep-dive explores why L-Menthol is the gold standard for flavoring, while its twin, D-Menthol, is a pale, often unpleasant, imitation.
To understand why L-Menthol and D-Menthol perform differently, we must first revisit the fundamental concept of chirality. Derived from the Greek word for “hand,” chirality refers to a molecule that lacks an internal plane of symmetry.
Imagine your hands. They are identical in structure—four fingers and a thumb—but they are mirror images of one another. No matter how you rotate or flip them, you cannot superimpose your right hand perfectly onto your left (palm to palm results in thumbs pointing in opposite directions). In chemistry, such molecules are called enantiomers.
Menthol (C10H20O) is a covalent compound containing a cyclohexane ring. What makes it fascinating to chemists is that it possesses three chiral centers (asymmetric carbon atoms). These are located at the 1, 2, and 5 positions of the ring. According to the 2n rule (where n is the number of chiral centers), menthol can theoretically exist in 23 = 8 different stereoisomeric forms.
These eight isomers are grouped into four pairs of enantiomers:
In nature, and specifically in the Mentha arvensis or Mentha piperita plants, the biological machinery of the plant is “programmed” to produce almost exclusively the (-)-Menthol isomer, also known as L-Menthol (Levorotatory).
The specific configuration of L-Menthol is (1R, 2S, 5R). This designation, based on the Cahn-Ingold-Prelog (CIP) priority rules, describes the exact spatial arrangement of the hydroxyl group (-OH), the methyl group (-CH3), and the isopropyl group (-CH(CH3)2) around the cyclohexane ring.
When we talk about the “cooling” sensation of menthol, we are specifically talking about the way this (1R, 2S, 5R) configuration interacts with human biology.
D-Menthol (or (+)-Menthol, with a (1S, 2R, 5S) configuration) is the mirror image of L-Menthol. While it has the same boiling point, the same density, and the same chemical reactivity in a vacuum, it behaves entirely differently when introduced to a biological system—like a vaper’s tongue and throat.
As noted by the American Chemical Society (ACS), “The human body is an inherently chiral environment. Our receptors, enzymes, and even our DNA are made of chiral building blocks (L-amino acids and D-sugars), meaning they can distinguish between enantiomers just as a right-handed glove distinguishes between a right and left hand” (Source: Journal of Chemical Education).
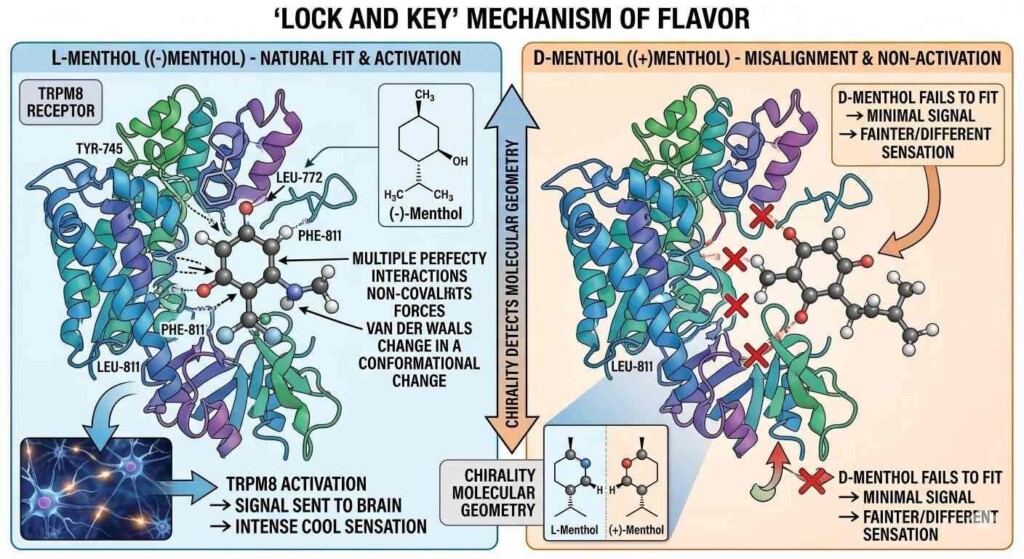
Lock & Key Diagram
The reason L-Menthol “tastes” cold isn’t because it lowers the temperature of your mouth. It is a chemical illusion. L-Menthol is an agonist for the TRPM8 (Transient Receptor Potential Melastatin 8) ion channel.
TRPM8 is a protein found in sensory neurons that is naturally activated by cold temperatures (typically below 26℃ / 79℉). When L-Menthol binds to this receptor, it lowers the threshold at which the channel opens. This allows sodium (Na+) and calcium (Ca2+) ions to flow into the cell, triggering an action potential that the brain interprets as “cold.”
Research published in Nature has demonstrated that the TRPM8 receptor is highly stereoselective. The binding pocket of the receptor is shaped specifically to accommodate the (1R, 2S, 5R) geometry of L-Menthol.
Furthermore, because D-Menthol doesn’t fit the “cooling” lock perfectly, it often ends up rattling around in other “locks”—specifically, receptors associated with bitterness or mustiness. This is why D-Menthol is frequently described as having a “medicinal,” “herbaceous,” or “musty” off-note that can ruin a delicate fruit or dessert e-liquid profile.
For an e-liquid manufacturer, understanding the nuances of the other isomers is vital for quality control. If your menthol source is “racemic” (a 50/50 mix of L and D) or contaminated with neomenthol, your flavor profile will suffer.
| Isomer | Common Name | Sensory Profile | Relative Cooling Potency |
| (1R,2S,5R) | L-Menthol | Fresh, clean, sharp peppermint, intense cooling. | 100% |
| (1S,2R,5S) | D-Menthol | Weak mint, musty, bitter, slight cooling. | ~5-10% |
| (1S,2S,5R) | (+)-Neomenthol | Musty, minty, slightly earthy. | <1% |
| (1R,2R,5S) | (-)-Neomenthol | Fresh, minty, but lacks cooling punch. | <1% |
| (1R,2S,5S) | (+)-Isomenthol | Camphoraceous, medicinal, woody. | <1% |
| (1S,2R,5R) | (-)-Isomenthol | Faint mint, mostly woody/earthy. | <1% |
| (1S,2S,5S) | (+)-Neoisomenthol | Very weak, slightly sweet/floral. | <1% |
| (1R,2R,5R) | (-)-Neoisomenthol | Musty, chemical, negligible cooling. | <1% |
As the table shows, if you aren’t using high-purity L-Menthol, you are essentially diluting your cooling effect with “chemical noise.” In the e-liquid industry, where “ice” flavors are expected to be crisp, these impurities lead to a “heavy” or “dirty” exhale that consumers dislike.
As a flavoring manufacturer, we often get asked: “Is natural L-Menthol better than synthetic?” The answer lies in the purity of the enantiomer.
Natural L-Menthol is extracted via the steam distillation of Mentha arvensis. The resulting peppermint oil is then chilled (dementholized), causing the L-Menthol to crystallize. Since the plant only makes the L-isomer, natural menthol is inherently “enantiopure.” However, natural sources can carry trace impurities from the plant, such as pulegone or menthofuran, which can alter the flavor profile.
The creation of synthetic L-Menthol was a milestone in industrial chemistry. For years, synthetic menthol was “racemic” (a mix of D and L), which was inferior for flavoring. However, in the 1980s, Ryoji Noyori developed a method for asymmetric catalysis, for which he was later awarded the Nobel Prize in Chemistry in 2001.
This process uses a chiral rhodium catalyst (specifically, Rh-BINAP) to ensure that the chemical reaction only produces the L-isomer. This allows manufacturers to produce “Nature-Identical” L-Menthol that is 99.9% pure, free from the “musty” D-isomer and the “earthy” plant impurities. (Source: NobelPrize.org).
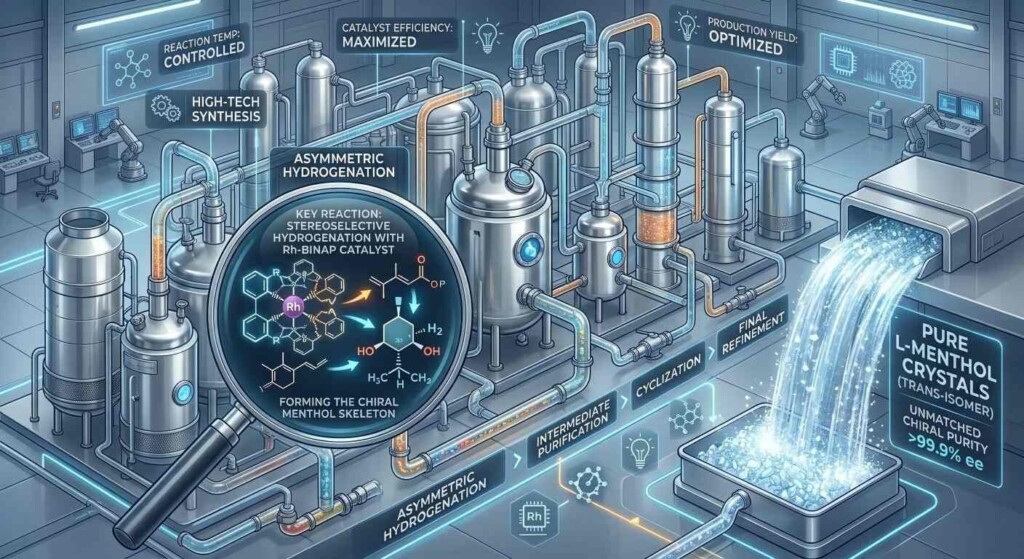
Takasago Infographic
In our laboratory, we have conducted extensive sensory panels on how chirality affects the final vape. The results are consistent: L-Menthol is non-negotiable for high-performance liquids.
One technical challenge in e-liquid manufacturing is the “crashing out” of menthol crystals. L-Menthol has a melting point of approximately 42℃ – 45℃. When formulated in high concentrations in high-VG (Vegetable Glycerin) liquids, L-Menthol can recrystallize if the temperature drops.
Interestingly, the presence of D-Menthol (as in a racemic mixture) can actually change the solubility and crystallization point. However, the trade-off in flavor quality is never worth the slight change in stability. We recommend using a high-purity L-Menthol dissolved in a PG (Propylene Glycol) carrier at a 10% or 20% “crushed menthol” solution to ensure long-term stability without compromising the “clean” hit.
E-liquid profiles often include sweeteners like Sucralose or Ethyl Maltol. L-Menthol interacts beautifully with these, as its “clean” profile allows the sweetness to shine. Conversely, the “musty” off-notes of D-Menthol or Isomenthol can react poorly with sweeteners, creating a flavor that tastes like “decaying vegetation” or “old medicine.”
To guarantee that our clients receive only the finest L-Menthol, we employ a multi-tiered analytical approach. You cannot tell the difference between L and D-Menthol by looking at them; they both appear as white, needle-like crystals.
Menthol is the most famous example, but it is far from the only one. The flavoring industry is a minefield of chiral traps:
When we develop a flavor concentrate—whether it’s a “Zesty Orange Ice” or a “Spearmint Blast”—we aren’t just mixing chemicals. We are curating a specific stereochemical profile to ensure that the user’s receptors are triggered in exactly the right way.
In the e-liquid market, “cheap” menthol is easy to find. Often, these are industrial-grade racemic mixtures intended for non-sensory applications (like topical ointments or industrial fresheners). Using these in a vape product is a recipe for brand failure.
A “medicinal” aftertaste is the number one reason consumers switch e-liquid brands. By insisting on validated L-Menthol, you are investing in:
To summarize, the difference between L-Menthol and D-Menthol is the difference between a key that fits the lock and a key that jams it.
As your manufacturing partner, we don’t just supply flavors; we supply the chemical certainty that your product will stand up to the most discerning palate.

Premium Product Shot
Are you struggling with a “medicinal” note in your menthol liquids? Or perhaps your “Ice” flavors aren’t delivering the punch your customers demand? Let’s talk science.
We offer comprehensive technical support for e-liquid brands looking to optimize their formulations. Whether you need a high-stability menthol solution or a custom-designed chiral flavor profile, our lab is at your disposal.
Contact us today to request:
| Contact Channel | Details |
| 🌐 Website: | www.cuiguai.com |
| 📧 Email: | info@cuiguai.com |
| ☎ Phone: | +86 0769 8838 0789 |
| 📱 WhatsApp: | +86 189 2926 7983 |
| 📍 Factory Address | Room 701, Building 3, No. 16, Binzhong South Road, Daojiao Town, Dongguan City, Guangdong Province, China |
The business scope includes licensed projects: food additive production. General projects: sales of food additives; manufacturing of daily chemical products; sales of daily chemical products; technical services, technology development, technical consultation, technology exchange, technology transfer, and technology promotion; biological feed research and development; industrial enzyme preparation research and development; cosmetics wholesale; domestic trading agency; sales of sanitary products and disposable medical supplies; retail of kitchenware, sanitary ware and daily sundries; sales of daily necessities; food sales (only sales of pre-packaged food).
Copyright ©Guangdong Unique Flavor Co., Ltd.All Rights Reserved. Privacy Policy