Author:R&D Team, CUIGUAI Flavoring
Published by:Guangdong Unique Flavor Co., Ltd.
Last Updated: May 08, 2026
WhatsApp & Telegram:+86 189 2926 7983

GC-MS E-Liquid Analysis
In the highly competitive landscape of international e-liquid manufacturing,flavor stability testingis the cornerstone of product quality and brand reputation. For business-to-business (B2B) flavor suppliers and manufacturers, ensuring that a volatile flavor profile remains consistent from the laboratory bench to the end consumer is a monumental scientific challenge. This challenge is exponentially magnified in markets subject to extreme climatic variations and rigorous logistics networks, such as the Russian Federation and the broader Eurasian Economic Union (EAEU).
Flavorings in e-liquids are not static; they are highly dynamic chemical systems. When aromatic compounds—ranging from light esters providing fruity top notes to heavy vanillin structures anchoring dessert profiles—are introduced into a matrix of Propylene Glycol (PG) and Vegetable Glycerin (VG), a cascade of molecular interactions begins. Over time, exposure to temperature fluctuations, oxygen, and ultraviolet radiation can induce oxidation, polymerization, and the formation of acetals, fundamentally altering the organoleptic properties of the product.
To meet the exacting demands of sophisticated consumers and stringent regulatory frameworks, manufacturers must employ robust flavor stability testing methods. This comprehensive guide explores the advanced analytical techniques required to guarantee product longevity, focusing onGas Chromatography-Mass Spectrometry (GC-MS), predictive thermodynamic models, and specialized methodologies tailored for complex global supply chains.
By investing in rigorous stability protocols, manufacturers can confidently expand their product lines. For those looking to explore a diverse range of thoroughly tested aromatic profiles, ourpremium flavor concentratesoffer a foundation for exceptional e-liquid formulations.
Before delving into the analytical instrumentation, it is imperative to understand the primary chemical pathways responsible for flavor degradation within an e-liquid matrix. A B2B manufacturer must anticipate these reactions during the formulation phase rather than merely reacting to them post-production.
Oxidation is the most ubiquitous threat to flavor stability. Terpenes, common in citrus and fruit profiles, are highly susceptible to oxidative degradation. For instance, d-limonene can readily oxidize into carvone or carveol, shifting a bright citrus note into a flat, piney, or even rancid off-note. This process is accelerated by heat and light, making light-proof packaging and nitrogen-flushing critical preventative measures.
Propylene Glycol (PG) is not merely an inert carrier; it is a reactive diol. When PG interacts with aldehydes—key flavor compounds found in vanilla, cherry, and almond profiles (e.g., benzaldehyde, vanillin)—acetalization occurs. PG acetals often have significantly higher boiling points and muted aromatic profiles compared to their parent aldehydes, leading to a phenomenon known as “flavor muting” over the product’s shelf life.
Dessert flavorings frequently contain reducing sugars and amino compounds. Even at room temperature, these can undergo slow Maillard reactions within the e-liquid, leading to a progressive darkening of the liquid (browning) and the development of caramel-like or burned off-notes. While sometimes desirable in tobacco or bakery profiles, uncontrolled Maillard reactions in fruit or clear liquids signify degradation.
At the heart of any sophisticated flavor laboratory is Gas Chromatography-Mass Spectrometry (GC-MS). This technique is universally recognized as the gold standard for analyzing volatile and semi-volatile compounds within complex matrices. For e-liquid flavor stability testing,GC-MS analysisis not optional; it is mandatory.
GC-MS operates through a two-stage process:
In stability testing, GC-MS analysis is utilized to establish a baseline chemical fingerprint of a freshly manufactured flavor concentrate or e-liquid. As the sample undergoes aging, subsequent GC-MS runs are compared against this baseline. Analysts look for:
To achieve reliable results, sample preparation is critical. E-liquids are heavily laden with PG and VG, which can overload standard GC columns and obscure trace flavor compounds. Techniques such as Solid-Phase Microextraction (SPME) or liquid-liquid extraction are often employed to isolate the aromatic volatiles from the heavy humectant base prior to injection. This ensures that the instrument detects subtle changes in the parts-per-million (ppm) range, which are vital for maintaining the delicate balance of top notes in premium e-liquids.
For further reading on how analytical chemistry integrates into large-scale production workflows, visit ourtechnical blog category.

E-Liquid Stability Testing
While GC-MS is exceptional for volatile compounds, a comprehensive stability testing protocol must account for non-volatiles, physical stability, and sensory perception.
HPLC is essential for analyzing non-volatile additives that do not readily vaporize in a GC inlet. This includes the quantification of active ingredients, certain complex sweeteners (like sucralose or steviol glycosides), and non-volatile bitter-masking agents. Monitoring the stability of these components is crucial, as the breakdown of sweeteners can drastically alter the pH and flavor profile of the liquid.
Advanced flavorings often utilize microencapsulation or complex emulsion technologies to protect volatile compounds or blend oil-soluble flavors into water/PG bases. Assessing emulsion stability involves measuring particle size distribution over time using dynamic light scattering (DLS). An increase in droplet size indicates coalescence, which precedes phase separation—a critical failure in e-liquid formulation.
Instruments cannot fully replicate the human palate. A trained sensory panel remains a vital component of stability testing. Sensory evaluation uses triangle tests and descriptive analysis to correlate the chemical changes detected by GC-MS analysis with actual human perception. If a GC-MS detects a 5% loss in a volatile ester, the sensory panel determines if this chemical variance crosses the threshold of consumer detection.
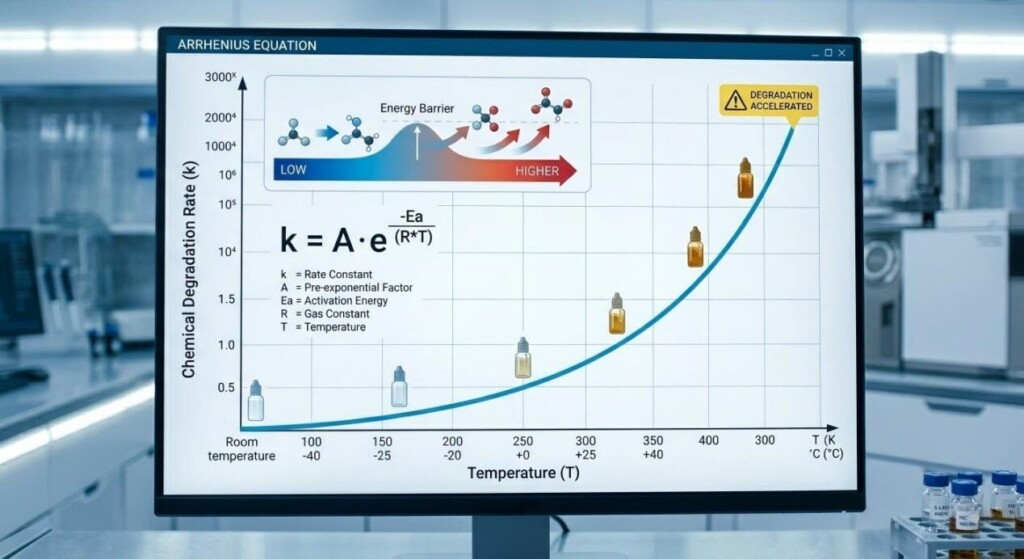
Waiting two years in real-time to determine if an e-liquid is stable is commercially unviable. Therefore, B2B manufacturers rely on accelerated aging protocols and thermodynamic modeling to generate accurateShelf Life Predictiondata within weeks.
Accelerated testing involves subjecting the e-liquid to elevated temperatures, humidity, and light exposure in specialized environmental chambers. The core principle relies on chemical kinetics: higher temperatures accelerate the rate of chemical reactions (such as oxidation and acetalization).
Shelf Life Prediction is mathematically grounded in the Arrhenius equation, which describes the temperature dependence of reaction rates. In the food and flavor industry, this is often simplified using the Q10temperature coefficient. The Q10factor represents the factor by which the reaction rate increases for every 10℃increase in temperature.
For many flavor degradation reactions in PG/VG matrices, the Q10value is approximately 2.0. This implies that storing a sample at 40℃(accelerated condition) rather than 20℃(standard room temperature) will cause reactions to occur four times faster (22). Therefore, three months of stability observed at 40℃can confidently predict twelve months of stability at 20℃.
While heat accelerates chemical degradation, extreme cold poses physical stability challenges. For e-liquids destined for the Russian market, where winter transit temperatures can plummet below -30℃, cold-shock testing is imperative. At low temperatures, the viscosity of VG increases exponentially, and certain flavor isolates may breach their solubility limits, leading to crystallization or precipitation. Shelf Life Prediction protocols for these regions must include freeze-thaw cycling to ensure the product remains a homogeneous solution upon returning to room temperature, without requiring the end-user to agitate the product.
Chemical Degradation Chart
To illustrate the practical application of these methods, consider the stabilization of a complex “Hami Melon & Vanilla Cream” e-liquid profile engineered for export.
Initial organoleptic panels reported that the profile lost its crisp melon top notes after three months of standard storage, while the vanilla aspect developed an undesirable, heavy syrupy characteristic.
The engineering team initiated a comprehensiveGC-MS Analysis. Comparing the degraded sample to the baseline chromatogram revealed two key issues:
Armed with this data, the formulation was adjusted. To combat oxidation, a trace amount of a natural antioxidant (tocopherol) was introduced into the matrix. To mitigate acetal formation, the ratio of the vanilla base was slightly adjusted, and a co-solvent technique utilizing a micro-emulsion was employed to sterically hinder the interaction between the vanillin molecules and the PG carrier.
The revised formulation underwent a 90-day accelerated stability program at 40℃, alongside freeze-thaw cycling to simulate transport through Siberian logistics routes. Post-test GC-MS analysis confirmed that ester depletion was halted, and vanillin acetalization was reduced to less than 2%, well below the sensory detection threshold. The resulting product achieved a validated 24-month shelf life, ensuring compliance and quality upon arrival in the Eurasian market.
Stability testing is not merely a quality control measure; it is a regulatory prerequisite. As global legislation surrounding vaping products tightens, verifiable stability data is essential for market access.
In the European Union, the Tobacco Products Directive (TPD) requires manufacturers to submit detailed toxicological and emissions data, which inherently relies on the stability of the liquid over its stated shelf life. Similarly, for the Russian and EAEU markets, compliance with GOST standards and Technical Regulations (TR CU) mandates rigorous safety and quality documentation. Demonstrating a scientifically backed shelf life through GC-MS and accelerated testing ensures that the product remains safe and chemically consistent with its original registration dossier, protecting the manufacturer from costly recalls and legal liabilities.
References to authoritative bodies such as theFlavor and Extract Manufacturers Association (FEMA)guidelines on GRAS status, and the analytical methodologies outlined by theAmerican Chemical Society (ACS), provide the framework for these necessary compliance protocols.
Mastering flavor stability is a complex but necessary endeavor for any serious B2B manufacturer in the e-liquid sector. By integrating rigorousGC-MS Analysis, holistic physical testing, and mathematically soundShelf Life Predictionmodels, manufacturers can engineer products that withstand the rigors of time and extreme global logistics. For markets with demanding consumers and challenging climates, this scientific rigor is the ultimate differentiator.

Product Quality & Logistics
Ensure your e-liquids stand the test of time and temperature. Partner with Cuiguai for rigorously tested, highly stable flavor concentrates engineered for global success.
Technical Consultation & Free Sample Request:Let our flavor chemists assist in your next formulation.
| Contact Channel | Details |
| 🌐 Website: | www.cuiguai.com |
| 📧 Email: | info@cuiguai.com |
| ☎ Phone: | +86 0769 8838 0789 |
| 📱 WhatsApp: | +86 189 2926 7983 |
| 📱Telegram: | +86 189 2926 7983 |
| 📍 Factory Address | Room 701, Building 3, No. 16, Binzhong South Road, Daojiao Town, Dongguan City, Guangdong Province, China |
O escopo de negócios inclui projetos licenciados: produção de aditivos alimentares. Projetos gerais: vendas de aditivos alimentares; fabricação de produtos químicos diários; vendas de produtos químicos diários; serviços técnicos, desenvolvimento de tecnologia, consultoria técnica, intercâmbio de tecnologia, transferência de tecnologia e promoção de tecnologia; pesquisa e desenvolvimento de rações biológicas; pesquisa e desenvolvimento de preparação enzimática industrial; atacado de cosméticos; agência de comércio doméstico; vendas de produtos sanitários e suprimentos médicos descartáveis; varejo de utensílios de cozinha, louças sanitárias e artigos diversos; vendas de necessidades diárias; vendas de alimentos (apenas vendas de alimentos pré-embalados).
Direitos autorais ©Guangdong Unique Flavor Co., Ltd.Todos os direitos reservados. Política de privacidade